What Happens When You Stop Taking Semaglutide?
Semaglutide for Weight Loss
Two-thirds regained
Participants in the STEP 1 extension regained two-thirds of their prior weight loss within one year of stopping semaglutide.
Wilding et al., Diabetes Obes Metab, 2022
Wilding et al., Diabetes Obes Metab, 2022
If you only read one thing
Most of the weight comes back. In the biggest study on this, people who stopped semaglutide regained about two-thirds of what they'd lost within a year. Their appetite went back to normal, and so did their blood pressure and cholesterol improvements. The drug works like blood pressure medication — it manages the problem while you take it, but it doesn't cure anything. Some people keep a small amount of weight off after stopping, but the majority of the benefit disappears.
Semaglutide produces substantial weight loss when used consistently. The STEP 1 trial showed 14.9% average body weight reduction over 68 weeks.[2] But what happens when patients stop? The extension of that same trial provides a clear, uncomfortable answer: most of the weight comes back. Within one year of discontinuation, participants regained two-thirds of what they had lost.[1]
That finding has reshaped the conversation around GLP-1 receptor agonists. Semaglutide works by mimicking a gut hormone that reduces appetite and slows gastric emptying. When the drug is removed, those effects disappear. Hunger returns. Portions increase. The metabolic adaptations that kept weight off reverse course. This is not a failure of willpower. It is the biology of obesity reasserting itself.
Key Takeaways
- Most of the weight comes back. In the biggest study on stopping semaglutide, people regained two-thirds of their weight loss within one year.
- Someone who dropped from 220 to 182 pounds would be back around 195 a year after stopping — some progress kept, most of it reversed.
- Blood pressure, cholesterol, and inflammation improvements also reverse. The drug manages these the way blood pressure medication does — while you take it.
- Semaglutide cuts how much food people eat by about 35% at a meal. When it's stopped, appetite returns to where it was.
- The weight coming back is not a willpower problem. It's your body's biology reasserting itself after the appetite-control signal stops.
- Keeping the weight off at two years is possible — but only for people who kept taking the drug. Nobody in the trials maintained 15%+ loss after stopping.
- About 40% of the weight lost on semaglutide is muscle, not fat. What you regain may not be the same tissue you lost.
The STEP 1 Extension: What the Landmark Data Shows
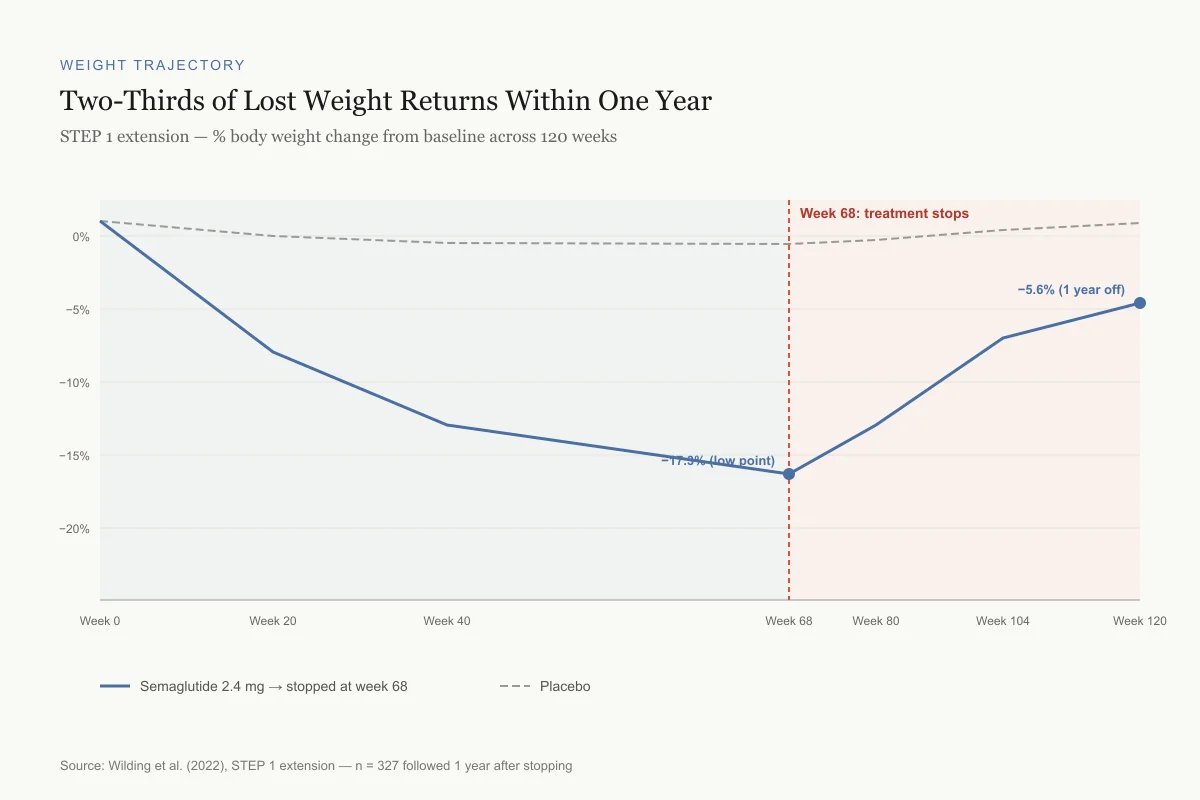
The clearest evidence on semaglutide discontinuation comes from the STEP 1 trial extension.[1] The original 68-week trial randomized 1,961 adults without diabetes to semaglutide 2.4 mg or placebo. At week 68, all treatments, including lifestyle intervention, were stopped. A subset of 327 participants was followed for an additional year.
The numbers tell a straightforward story. Semaglutide participants lost an average of 17.3% of body weight by week 68. After discontinuation, they regained 11.6 percentage points by week 120, ending up with a net loss of just 5.6% from their starting weight. Placebo participants, who had lost 2.0%, regained 1.9 percentage points.
The cardiometabolic improvements followed the same pattern. Reductions in waist circumference, blood pressure, C-reactive protein, and lipid profiles all reverted toward baseline after semaglutide was withdrawn.[1] The metabolic benefits were not permanent changes. They required ongoing treatment.
Weight Trajectory
Two-Thirds of Lost Weight Returns Within One Year
STEP 1 extension — % body weight change from baseline across 120 weeks
Source: Wilding et al. (2022), STEP 1 extension — n = 327 followed 1 year after stopping
 View as image
View as imageSTEP 4: Continued Treatment vs. Withdrawal
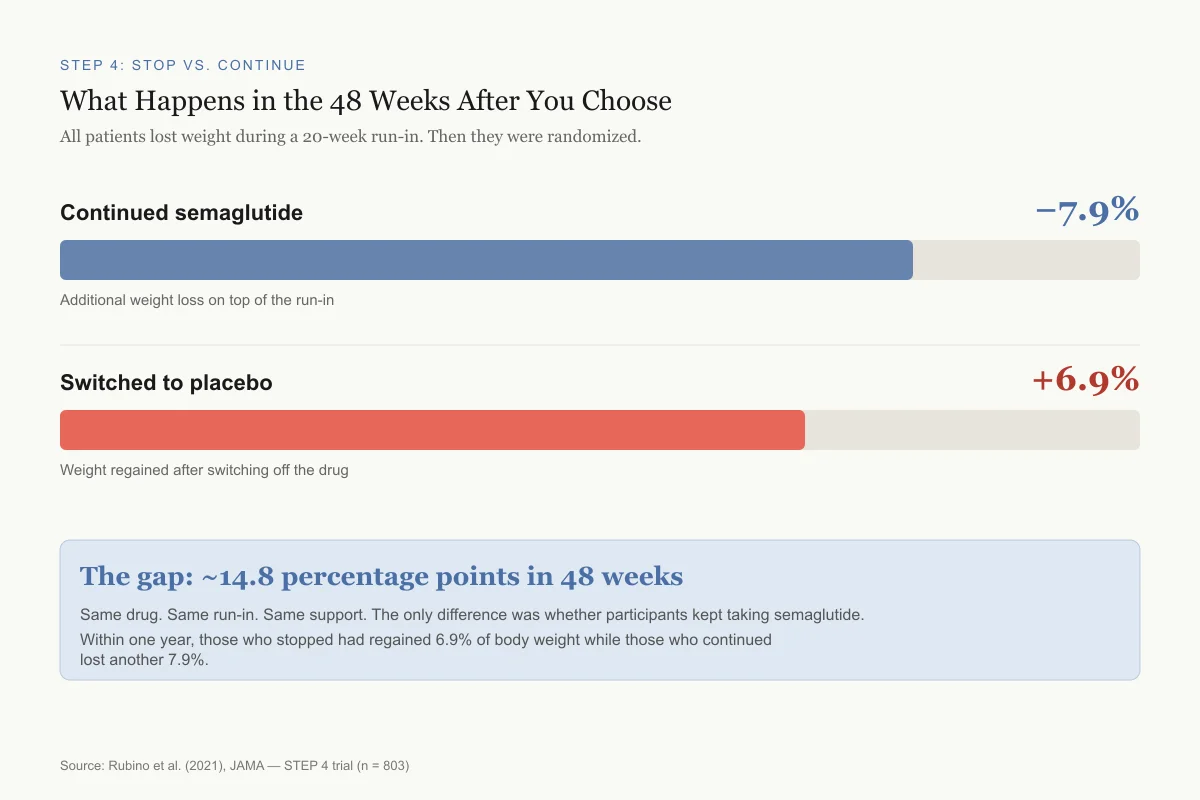
The STEP 4 trial was designed specifically to answer the continuation question.[3] All 902 participants received semaglutide during a 20-week run-in period. Those who reached the maintenance dose were then randomized: two-thirds continued semaglutide, one-third switched to placebo.
The divergence was immediate and sustained. Participants who continued semaglutide lost an additional 7.9% of body weight from week 20 to week 68. Those switched to placebo regained 6.9% over the same period.[3] By the end of the trial, the gap between groups was approximately 14.8 percentage points.
This trial design was particularly revealing because every participant had already experienced the drug's effects during run-in. The withdrawal group did not lose their memory of what eating on semaglutide felt like. Their bodies simply reverted to pre-treatment appetite and metabolic patterns once the pharmacological signal was removed.
STEP 4: Stop vs. Continue
What Happens in the 48 Weeks After You Choose
All patients lost weight on semaglutide during a 20-week run-in. Then they were randomized.
Continued semaglutide
-7.9%
Additional weight loss on top of the run-in
Switched to placebo
+6.9%
Weight regained after switching off the drug
The gap: ~14.8 percentage points in 48 weeks
Same drug, same run-in, same support. The only difference was whether participants kept taking semaglutide. Within one year, those who stopped had regained 6.9% while those who continued lost another 7.9%.
Source: Rubino et al. (2021), JAMA — STEP 4 trial (n = 803)
 View as image
View as imageWhy Appetite Returns
Semaglutide reduces food intake through multiple mechanisms: it slows gastric emptying, enhances satiety signaling, and acts on brain regions that control appetite and food reward. Gibbons et al. (2021) quantified these effects in a crossover trial with 15 participants with type 2 diabetes.[6]
After 12 weeks of oral semaglutide, ad libitum energy intake at lunch decreased by approximately 35% (949 kJ less than placebo). Participants reported less hunger, fewer food cravings, and a lower desire to eat. Their preference for high-fat foods decreased.
These appetite effects are drug-dependent. When treatment ends, the GLP-1 receptor stimulation that produced them disappears. Endogenous GLP-1 secretion returns to its baseline level, which in people with obesity is often insufficient to counteract the drive to eat. The result: caloric intake climbs back toward pre-treatment levels. This is the primary driver of weight regain.
Understanding how semaglutide works at the receptor level clarifies why stopping produces such consistent regain. The drug's half-life is approximately one week. Within a few weeks of the last injection, plasma concentrations drop below therapeutic levels, and the appetite-suppressing effects fade.
What Happens to Body Composition
Weight regain after semaglutide discontinuation raises specific concerns about body composition. Bikou et al. (2024) conducted a systematic review of semaglutide's effects on lean mass across clinical trials.[7] They found that approximately 39% of total weight lost on semaglutide came from lean body mass, with the remaining 61% from fat mass.
This ratio matters for what happens after stopping. When weight is regained, the composition of that regain tends to favor fat over lean tissue. A person who loses 15 kg (with 5.8 kg from muscle and 9.2 kg from fat) and then regains 10 kg may end up with more fat and less muscle than they started with, even though their scale weight is only 5 kg below baseline.
The STEP program analyses reported by O'Neil and Rubino (2022) showed that semaglutide did improve body composition overall, reducing waist circumference and visceral fat disproportionately.[8] But the post-discontinuation body composition trajectory has not been studied with the same rigor. Whether regained weight has the same fat-to-lean ratio as the original loss remains an open question.
What Stays the Same on Continued Treatment
If discontinuation leads to regain, what does continued treatment look like? The STEP 5 trial provides the best long-term data.[4] Over 104 weeks of continued semaglutide 2.4 mg, participants maintained a mean weight loss of 15.2%. The placebo group lost 2.6%.
Two years of data showed no significant plateau-related rebound. Weight loss stabilized between weeks 60 and 104 rather than reversing. This suggests that semaglutide's efficacy is durable as long as treatment continues, answering one concern that the body might "adapt" and stop responding.
For cardiovascular risk, the SELECT trial (Lincoff et al., 2023) demonstrated that semaglutide reduced major adverse cardiovascular events by 20% in people with obesity and established cardiovascular disease, even without diabetes.[5] Whether those cardiovascular benefits persist after discontinuation has not been tested in a dedicated trial, but the STEP 1 extension data showing cardiometabolic marker reversion suggests they likely do not.
Real-World Discontinuation Looks Different
Clinical trial participants are a selected population. Real-world discontinuation patterns are messier and sometimes more optimistic. Del Prete et al. (2025) followed 175 patients with type 2 diabetes who used semaglutide in routine clinical practice.[9]
Their findings showed that real-world weight maintenance after semaglutide varies considerably. Many patients restart treatment after a gap, switch to alternative GLP-1 drugs, or combine semaglutide with other interventions. The clean "on-drug then off-drug" trajectory seen in controlled trials rarely plays out in clinical practice.
A 2026 Cleveland Clinic analysis of nearly 8,000 patients found that those who stopped semaglutide or tirzepatide regained an average of only 0.5% body weight at one year, far less than the clinical trial data predicted. The likely explanation: many patients in the real world do not stay off treatment permanently. They cycle through periods of use, creating a pattern that trials are not designed to capture.
This does not mean discontinuation is harmless. It means that the binary framing of "on semaglutide" versus "off semaglutide" does not reflect how the drug is actually used.
The "Forever Drug" Question
The consistency of weight regain after discontinuation has led to semaglutide being described as a "forever drug," one that must be taken indefinitely to maintain its effects. The clinical evidence supports this framing. Obesity is a chronic disease with biological drivers (appetite regulation, metabolic set points, hormonal signaling) that persist regardless of prior weight loss.
This parallels how other chronic conditions are managed pharmacologically. Blood pressure medications do not cure hypertension; they manage it. Statins do not permanently lower cholesterol; they suppress its production for as long as the drug is taken. Semaglutide appears to work the same way for appetite and body weight regulation. The STEP 5 data showing sustained 15.2% weight loss at two years with no loss of efficacy supports this chronic-use model.[4]
From a dosing perspective, some research is exploring whether lower maintenance doses might sustain benefits with fewer side effects and lower cost. The original STEP trials used a fixed 2.4 mg weekly dose, but real-world clinicians sometimes reduce doses after patients reach their target weight.
Whether dose reduction leads to partial regain is not well studied. The STEP 4 data shows that full discontinuation clearly produces substantial regain.[3] Whether going from 2.4 mg to 1.0 mg weekly preserves most of the benefit is a clinical question without a definitive trial answer. The cost implications are substantial: at list prices exceeding $1,000 per month in the United States, indefinite treatment represents a significant financial commitment that insurance coverage does not always address.
How Stopping Differs From Never Starting
One nuance frequently lost in the discontinuation discussion is that people who stop semaglutide after a period of weight loss are not in the same position as if they had never taken the drug. Even after regaining two-thirds of lost weight, STEP 1 extension participants retained a net 5.6% weight loss from baseline.[1] A 5.6% sustained reduction in body weight is clinically meaningful. It falls within the range associated with improvements in blood pressure, glycemic control, and liver fat.
The question is whether those retained benefits justify a temporary course of treatment. For some patients, the answer may be yes: a period of semaglutide use that produces 17% weight loss, followed by partial regain to a 5-6% net loss, still leaves them better off than their starting point. For others, particularly those whose cardiovascular risk reduction depends on maintaining larger weight losses, temporary treatment may be insufficient.
The STEP program did not design trials to answer this question directly. The discontinuation extension was exploratory, and participants who stopped semaglutide also stopped the lifestyle intervention component. Whether continued exercise and dietary modification after drug withdrawal could slow the regain trajectory remains unstudied in this specific trial context.
What the Evidence Does Not Tell Us
Several gaps exist in the discontinuation research. The STEP 1 extension followed participants for one year after stopping. Whether regain continues beyond that point, stabilizes at two-thirds, or eventually returns to baseline is unknown. The Oxford meta-analysis from early 2026 projected return to baseline at approximately 18 months, but projections are not the same as measured outcomes.
The effect of structured exercise during and after discontinuation has not been isolated in a semaglutide-specific trial. Exercise may help preserve lean mass and mitigate regain, but the magnitude of that effect relative to the biological drive for weight regain is uncertain.
Long-term psychological effects of the regain cycle, stopping a drug that reduced appetite and receiving the signal that weight management requires permanent pharmacotherapy, have not been systematically studied. The side effect profile of long-term use beyond two years also has limited data.
The Bottom Line
Stopping semaglutide produces consistent, substantial weight regain driven by the return of pre-treatment appetite and metabolic patterns. The STEP 1 extension showed two-thirds of lost weight returning within one year. The STEP 4 trial confirmed that continued treatment prevents this regain while discontinuation reverses it. Real-world data shows more variability, partly because many patients restart treatment. The evidence frames semaglutide as a chronic treatment for a chronic disease, not a short-term intervention with lasting effects.
Sources & References
- 1RPEP-06592·Wilding, John P H et al. (2022). “Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension..” Diabetes.Study breakdown →PubMed →↩
- 2RPEP-05873·Wilding, John P H et al. (2021). “Once-Weekly Semaglutide in Adults with Overweight or Obesity..” The New England journal of medicine.Study breakdown →PubMed →↩
- 3RPEP-05731·Rubino, Domenica et al. (2021). “What Happens When You Stop Semaglutide? The STEP 4 Weight Regain Study.” JAMA.Study breakdown →PubMed →↩
- 4RPEP-06130·Garvey, W Timothy et al. (2022). “Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial..” Nature medicine.Study breakdown →PubMed →↩
- 5RPEP-07120·Lincoff, A Michael et al. (2023). “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes..” The New England journal of medicine.Study breakdown →PubMed →↩
- 6RPEP-05404·Gibbons, Catherine et al. (2021). “Oral Semaglutide Cuts Daily Food Intake by 39% and Improves Eating Control.” Diabetes.Study breakdown →PubMed →↩
- 7RPEP-07858·Bikou, Alexia et al. (2024). “A systematic review of the effect of semaglutide on lean mass: insights from clinical trials..” Expert opinion on pharmacotherapy.Study breakdown →PubMed →↩
- 8RPEP-06403·O'Neil, Patrick M et al. (2022). “Exploring the wider benefits of semaglutide treatment in obesity: insight from the STEP program..” Postgraduate medicine.Study breakdown →PubMed →↩
- 9RPEP-10680·Del Prete, Michela et al. (2025). “Semaglutide Maintains Weight Loss Through 24 Months in Real-World Diabetic Patients — Injectable and Oral Forms Both Effective.” Eating and weight disorders : EWD.Study breakdown →PubMed →↩