Tirzepatide vs Semaglutide: Which Produces More Weight Loss?
Tirzepatide Research
20.2% vs 13.7%
In the SURMOUNT-5 head-to-head trial, tirzepatide produced 20.2% mean weight loss at 72 weeks compared to 13.7% for semaglutide in adults with obesity.
Aronne et al., NEJM, 2025
Aronne et al., NEJM, 2025
If you only read one thing
Tirzepatide wins on weight loss. In the first head-to-head trial (SURMOUNT-5, 2025), tirzepatide users lost 20.2% of their body weight over 72 weeks compared to 13.7% on semaglutide — roughly 50 pounds vs 33 pounds on average. About 1 in 3 tirzepatide users dropped 25% or more of their body weight, compared to about 1 in 6 semaglutide users. Surprisingly, tirzepatide also had fewer GI dropouts (2.7% vs 5.6%). The catch: semaglutide has proven cardiovascular benefits (it reduces heart attacks and strokes in people with obesity plus heart disease), and tirzepatide doesn't have that data yet. For pure weight loss, tirzepatide is the stronger choice; for someone with heart disease already, semaglutide's track record still matters.
For years, the question of whether tirzepatide or semaglutide produces greater weight loss could only be answered indirectly, by comparing results across different trials with different patient populations. That changed in 2025, when the SURMOUNT-5 trial published the first head-to-head comparison in the New England Journal of Medicine. The answer was clear: tirzepatide produced significantly more weight loss than semaglutide at 72 weeks, with a better gastrointestinal tolerability profile. Understanding how tirzepatide's dual mechanism differs from semaglutide's single GLP-1 receptor agonism helps explain why, and the SURMOUNT trial program provides the full context for tirzepatide's body composition effects.
Key Takeaways
- Tirzepatide wins on pure weight loss. The first head-to-head trial showed 50 pounds lost on average, versus 33 pounds on semaglutide, over 72 weeks.
- About 1 in 3 people on tirzepatide dropped 25% or more of their body weight. On semaglutide, it was closer to 1 in 6.
- Surprising twist: tirzepatide also had fewer people quitting from side effects. Usually the stronger drug is rougher. Here it was gentler.
- The difference comes from biology. Tirzepatide hits two hormone targets (GLP-1 and GIP). Semaglutide only hits one.
- Semaglutide has something tirzepatide doesn't: proven reduction in heart attacks and strokes in people with heart disease.
- For someone who just wants to lose weight, tirzepatide is now the stronger choice on every major measure.
- For someone who already has heart disease, semaglutide's longer track record still matters until tirzepatide's heart-outcomes trial reports.
SURMOUNT-5: The Head-to-Head Answer
The SURMOUNT-5 trial (Aronne et al., 2025) was a Phase 3b, open-label, controlled trial that randomized 751 adults with obesity but without type 2 diabetes to receive the maximum tolerated dose of either tirzepatide (10 mg or 15 mg) or semaglutide (1.7 mg or 2.4 mg) subcutaneously once weekly for 72 weeks.[1]
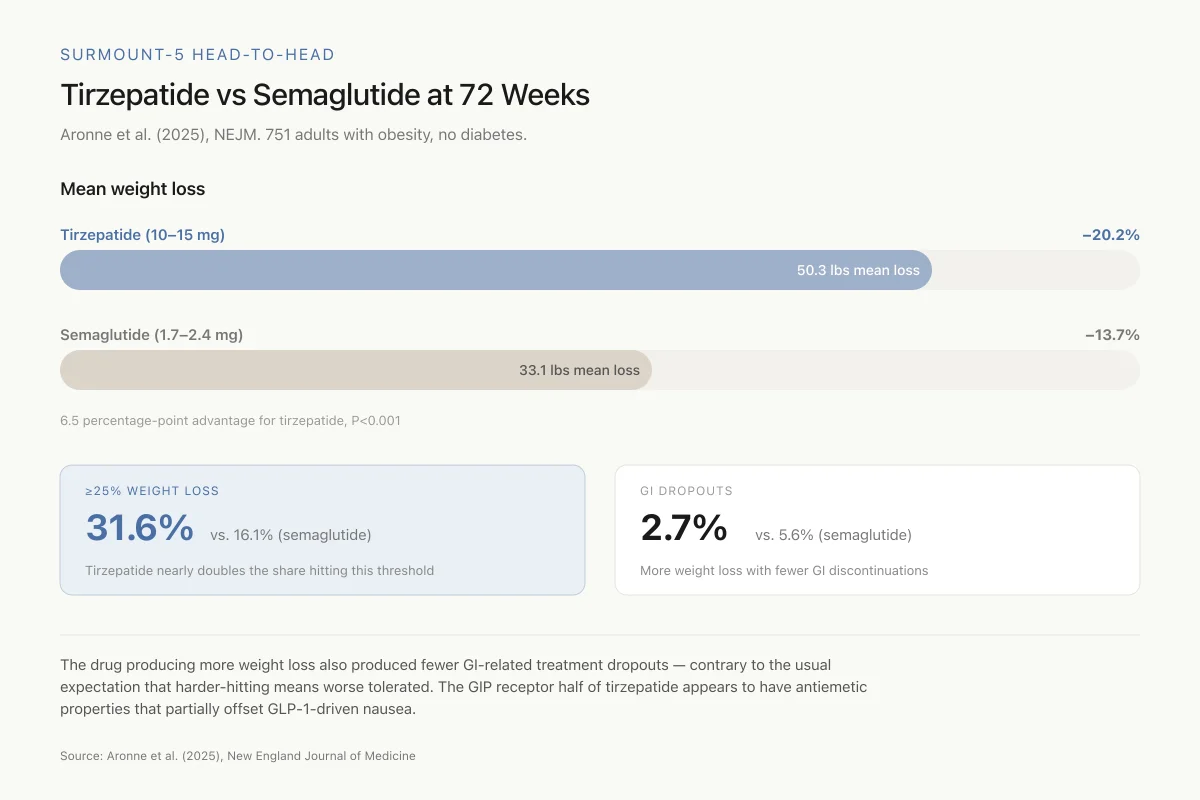
The primary endpoint was percent change in body weight at week 72. Tirzepatide produced a least-squares mean weight loss of 20.2% (95% CI, -21.4 to -19.1) compared to 13.7% (95% CI, -14.9 to -12.6) for semaglutide. The estimated treatment difference of 6.5 percentage points was statistically significant (P<0.001).
The secondary endpoints reinforced the primary finding. In absolute terms, tirzepatide users lost a mean of 50.3 lbs compared to 33.1 lbs for semaglutide users. For waist circumference, the change was -18.4 cm with tirzepatide versus -13.0 cm with semaglutide (P<0.001). Among tirzepatide users, 31.6% achieved at least 25% weight loss compared to 16.1% on semaglutide. Tirzepatide outperformed semaglutide on all five key secondary endpoints.
The safety profile favored tirzepatide on gastrointestinal tolerability. GI adverse events leading to treatment discontinuation occurred in 2.7% of tirzepatide users versus 5.6% of semaglutide users. This finding, that the drug producing more weight loss also produced fewer GI-related dropouts, was unexpected given the general assumption that more aggressive weight loss comes with more side effects.
SURMOUNT-5 Head-to-Head
Tirzepatide vs Semaglutide at 72 Weeks
Aronne et al. (2025), NEJM. 751 adults with obesity, no diabetes.
6.5 percentage-point advantage for tirzepatide, P<0.001
The drug producing more weight loss also produced fewer GI-related treatment dropouts — contrary to the usual expectation that harder-hitting means worse tolerated. The GIP receptor half of tirzepatide appears to have antiemetic properties that partially offset GLP-1-driven nausea.
Source: Aronne et al. (2025), New England Journal of Medicine
 View as image
View as imageWhy SURMOUNT-5 Has Limitations
SURMOUNT-5 was an open-label trial. Both patients and investigators knew which drug each participant received. This matters because weight loss outcomes can be influenced by expectation effects, dietary and exercise behavior, and reporting bias. Double-blind trials, where neither party knows the treatment assignment, are the gold standard for eliminating these confounders. The open-label design does not invalidate the results, but it means the 6.5 percentage point difference should be interpreted as a ceiling estimate rather than a precise number.
The trial also compared maximum tolerated doses rather than fixed doses. Tirzepatide was dosed at 10 mg or 15 mg; semaglutide was dosed at 1.7 mg or 2.4 mg. The 2.4 mg semaglutide dose is the highest available for obesity (Wegovy), but the semaglutide dose range tested did not include the experimental higher doses being studied in ongoing trials. Future semaglutide formulations may narrow the gap.
The study population was limited to adults with obesity but without type 2 diabetes. Whether the same magnitude of difference holds in diabetic populations, where the drugs' glucose-lowering effects interact with weight loss, is addressed by separate data.
Before SURMOUNT-5: Indirect Comparisons
Before the head-to-head trial, researchers used indirect comparisons and real-world data to estimate the difference between the two drugs.
SURPASS-2 (Type 2 Diabetes): Frias et al. (2021) published the first randomized comparison of tirzepatide and semaglutide, though in patients with type 2 diabetes rather than obesity.[2] In this 40-week trial, 1,879 patients were randomized to tirzepatide (5, 10, or 15 mg) or semaglutide (1 mg). Tirzepatide 15 mg reduced HbA1c by 2.46% compared to 1.86% for semaglutide 1 mg. Weight loss at 40 weeks was 12.4 kg with tirzepatide 15 mg versus 6.2 kg with semaglutide 1 mg.
A critical caveat: SURPASS-2 compared tirzepatide's maximum dose against semaglutide's 1 mg dose, which was the highest approved dose for diabetes at the time. The 2.4 mg obesity dose of semaglutide (Wegovy) was not included. This dose asymmetry made the comparison favorable to tirzepatide by design, and clinicians who cited SURPASS-2 as evidence that tirzepatide was "twice as effective" as semaglutide were drawing an invalid cross-dose comparison.
JAMA Real-World Data: Rodriguez et al. (2024) analyzed electronic health records of 18,386 adults with overweight or obesity who received either tirzepatide or semaglutide labeled for type 2 diabetes.[3] At 12 months, tirzepatide users lost 5.9 percentage points more body weight than semaglutide users. This real-world difference was smaller than the 6.5 point difference in SURMOUNT-5, but the finding is notable because real-world studies capture patient behavior and adherence patterns that clinical trials cannot.
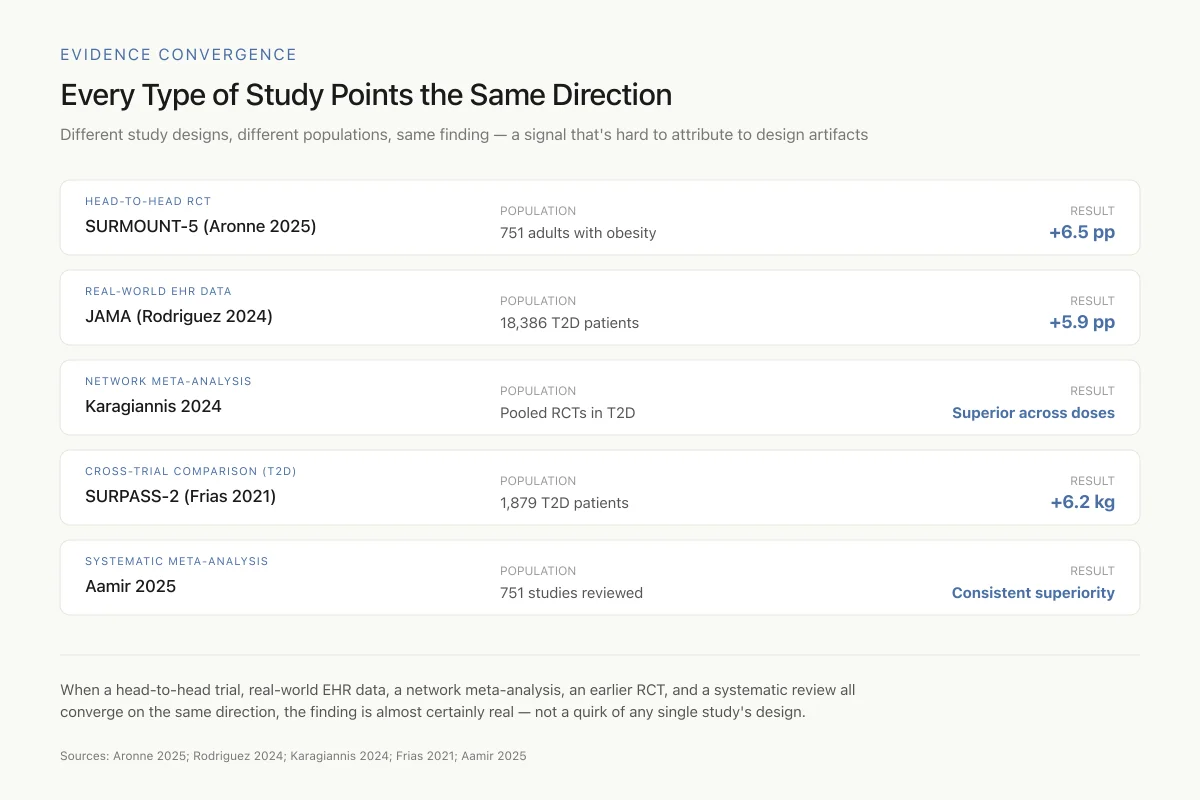
Network Meta-Analysis: Karagiannis et al. (2024) conducted a systematic review and network meta-analysis of randomized controlled trials comparing the two drugs in type 2 diabetes.[4] When comparing at matched doses and accounting for differences in trial design, tirzepatide showed superior weight loss and glycemic control across all dose comparisons.
Comprehensive Meta-Analysis: Aamir et al. (2025) synthesized both clinical trial data and real-world evidence in a meta-analysis that included 751 studies identified through systematic search, with a final included set comparing tirzepatide and semaglutide for weight reduction.[5] The analysis confirmed tirzepatide's superiority for weight loss across both controlled trial and real-world settings.
Evidence Convergence
Every Type of Study Points the Same Direction
Different study designs, different populations, same finding — a signal that's hard to attribute to design artifacts
When a head-to-head trial, real-world EHR data, a network meta-analysis, an earlier RCT, and a systematic review all converge on the same direction, the finding is almost certainly real — not a quirk of any single study's design.
Sources: Aronne 2025; Rodriguez 2024; Karagiannis 2024; Frias 2021; Aamir 2025
 View as image
View as imageThe GI Tolerability Difference
One of the most practically relevant differences between tirzepatide and semaglutide involves gastrointestinal side effects. Nausea, vomiting, and diarrhea are the most common adverse events with both drugs and the primary reason patients discontinue treatment.
Borner et al. (2025) investigated this difference in preclinical models and found that tirzepatide's GIP receptor agonism appears to have antiemetic properties that partially offset the nausea induced by GLP-1 receptor activation.[6] In animal models, tirzepatide produced equivalent food intake reduction and body weight loss with fewer GI adverse events compared to semaglutide. The study provided mechanistic evidence for what SURMOUNT-5 observed clinically: tirzepatide's dual mechanism may produce a more tolerable side effect profile than pure GLP-1 agonism.
Safwan et al. (2025) analyzed gastrointestinal safety data for semaglutide and tirzepatide versus placebo across clinical trials in obese individuals.[7] Both drugs increased GI adverse events compared to placebo, but the severity and discontinuation rates differed between the two compounds.
Fahim et al. (2025) conducted a broader comparative safety analysis examining side effect profiles beyond GI symptoms.[8] The analysis found that while both drugs share a similar overall safety profile, the specific pattern of adverse events differs in ways that may influence treatment selection for individual patients. Both drugs carry warnings for pancreatitis, gallbladder events, and thyroid C-cell tumors (based on rodent data). The clinical significance of these warnings differs between the drugs, and ongoing pharmacovigilance continues to refine the safety picture as millions of patients use these medications in real-world settings.
The STEP vs SURMOUNT Context
The SURMOUNT-5 comparison is best understood alongside the individual trial programs for each drug.
Semaglutide (STEP program): Wilding et al. (2021) published the STEP 1 trial showing that semaglutide 2.4 mg produced 14.9% weight loss at 68 weeks compared to 2.4% with placebo in adults with obesity.[9] This established semaglutide as a landmark obesity treatment, roughly tripling the weight loss achieved by prior medications.
Tirzepatide (SURMOUNT program): The SURMOUNT-1 trial showed tirzepatide 15 mg produced 22.5% weight loss at 72 weeks versus 3.1% with placebo, a result that exceeded semaglutide's STEP 1 data by approximately 7.6 percentage points. However, cross-trial comparisons are unreliable because the patient populations, trial durations, and background lifestyle interventions differ.
SURMOUNT-5 resolved this by testing both drugs in the same trial under the same conditions. The result, a 6.5 percentage point advantage for tirzepatide, was consistent with what the cross-trial comparisons had suggested but smaller than the crude difference between SURMOUNT-1 and STEP 1 implied.
The convergence of results across trial types strengthens confidence in the finding. When a head-to-head trial, multiple real-world studies, indirect comparisons, and meta-analyses all point in the same direction, the signal is likely real rather than an artifact of trial design. The consistency also suggests that the advantage is robust across different patient populations, dosing regimens, and healthcare settings.
What the Data Does Not Answer
Several clinically important questions remain unresolved even after SURMOUNT-5.
Long-term outcomes. SURMOUNT-5 followed patients for 72 weeks. Whether tirzepatide's superiority persists at 2, 5, or 10 years is unknown. Weight loss maintenance after discontinuation of either drug is poor, with studies showing rapid weight regain once treatment stops.
Cardiovascular outcomes. Semaglutide has demonstrated cardiovascular benefit in the SELECT trial (reduced major adverse cardiovascular events in patients with obesity and established cardiovascular disease). Tirzepatide does not yet have a completed cardiovascular outcomes trial. Greater weight loss does not automatically translate to better cardiovascular outcomes.
Body composition. Both drugs cause loss of lean mass alongside fat mass. Whether the ratio of fat to lean mass loss differs between the two drugs is an active area of research. Tirzepatide's body composition effects are being studied in post-hoc analyses of SURMOUNT trial data.
Cost-effectiveness. Both drugs are expensive. Whether tirzepatide's greater weight loss justifies its pricing relative to semaglutide depends on how payors value incremental weight loss and how drug pricing evolves. Mounjaro and Zepbound are tirzepatide's two brand names for diabetes and obesity, respectively.
Head-to-head with resistance exercise. Neither SURMOUNT-5 nor any existing trial has compared tirzepatide versus semaglutide when both groups also perform structured resistance exercise to preserve lean mass. This is a practical gap because most clinical guidelines now recommend exercise during GLP-1 agonist therapy.
Switching between drugs. Patients who do not tolerate or respond adequately to semaglutide may switch to tirzepatide, and vice versa. No controlled trial has studied the outcomes of switching between these drugs, though real-world data on this question is beginning to emerge.
Retatrutide and beyond. Eli Lilly's triple agonist retatrutide (GLP-1/GIP/glucagon) has shown even greater weight loss than tirzepatide in Phase II data. The comparison between tirzepatide and semaglutide may become less clinically relevant as newer multi-agonist drugs enter the market.
Safety
ModerateWeight loss ≠ cardiovascular benefit — don't assume tirzepatide wins that race too
Concern
Because tirzepatide produces more weight loss, it's tempting to assume it will also reduce heart attacks and strokes more than semaglutide. That assumption is not supported by current data. Semaglutide has the SELECT trial showing a 20% reduction in major adverse cardiovascular events in obesity with heart disease. Tirzepatide's equivalent trial (SURMOUNT-MMO) is still running.
What the research says
Until tirzepatide's cardiovascular outcomes data lands, the drug with more weight loss is not automatically the drug with more cardiovascular benefit. For patients with established cardiovascular disease, semaglutide currently has the proven endpoint.
Particularly relevant for: Patients with obesity plus atherosclerotic cardiovascular disease, prior heart attack, or prior stroke
What to do
If your primary goal is weight loss, tirzepatide is the stronger pick. If your primary goal is preventing a second cardiovascular event, semaglutide currently has the trial data. Re-evaluate when SURMOUNT-MMO reads out.
SELECT trial (Lincoff 2023); SURMOUNT-MMO (ongoing); Aronne 2025
The Bottom Line
The SURMOUNT-5 head-to-head trial showed tirzepatide produces significantly more weight loss than semaglutide (20.2% vs 13.7% at 72 weeks) with fewer GI-related treatment discontinuations (2.7% vs 5.6%). This finding is consistent across real-world data (Rodriguez et al., 2024) and meta-analyses (Karagiannis et al., 2024; Aamir et al., 2025). The GIP receptor component of tirzepatide's dual mechanism may explain both the greater weight loss and the better GI tolerability. Open questions include long-term outcomes, cardiovascular benefits, body composition effects, and cost-effectiveness.
Sources & References
- 1RPEP-09982·Aronne, Louis J et al. (2025). “Tirzepatide vs Semaglutide for Obesity: The SURMOUNT-5 Head-to-Head Trial.” The New England journal of medicine.Study breakdown →PubMed →↩
- 2RPEP-05391·Frías, Juan P et al. (2021). “SURPASS-2: Tirzepatide Beats Semaglutide for Blood Sugar and Weight Loss in Head-to-Head Trial.” The New England journal of medicine.Study breakdown →PubMed →↩
- 3RPEP-09163·Rodriguez, Patricia J et al. (2024). “Tirzepatide vs Semaglutide for Weight Loss: First Real-World Head-to-Head Comparison.” JAMA internal medicine.Study breakdown →PubMed →↩
- 4RPEP-08519·Karagiannis, Thomas et al. (2024). “Subcutaneously administered tirzepatide vs semaglutide for adults with type 2 diabetes: a systematic review and network meta-analysis of randomised controlled trials..” Diabetologia.Study breakdown →PubMed →↩
- 5RPEP-09726·Aamir, Ahmad Bin et al. (2025). “Tirzepatide vs Semaglutide for Weight Loss: Head-to-Head Meta-Analysis Including Real-World Data.” Journal of clinical medicine research.Study breakdown →PubMed →↩
- 6RPEP-10204·Borner, Tito et al. (2025). “Why Tirzepatide Causes Less Nausea Than Semaglutide: The GIP Component Acts as a Built-In Anti-Nausea Agent.” Science advances.Study breakdown →PubMed →↩
- 7RPEP-13360·Safwan, Moaz et al. (2025). “Gastrointestinal safety of semaglutide and tirzepatide vs. placebo in obese individuals without diabetes: a systematic review and meta analysis..” Annals of Saudi medicine.Study breakdown →PubMed →↩
- 8RPEP-10881·Fahim, Sally A et al. (2025). “Tirzepatide May Be Safer Than Semaglutide for Obesity Treatment, With Fewer Side Effects and Better Bone and Kidney Protection.” Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie.Study breakdown →PubMed →↩
- 9RPEP-05873·Wilding, John P H et al. (2021). “Once-Weekly Semaglutide in Adults with Overweight or Obesity..” The New England journal of medicine.Study breakdown →PubMed →↩