BPC-157 and the Heart: What Animal Studies Show
Peptides and Cardiac Repair
Zero Human Cardiac Trials
BPC-157 has demonstrated cardioprotective effects across dozens of animal studies, reversing arrhythmias, reducing myocardial infarct size, and restoring heart function in rats and mice. Not one of these findings has been tested in a human cardiovascular trial.
Sikiric et al., Biomedicines, 2022
Sikiric et al., Biomedicines, 2022
If you only read one thing
In rat studies, BPC-157 has done a lot of cardiovascular things: reduced heart attack damage, reversed chemo-induced heart failure, stopped drug-triggered arrhythmias, and normalized blood pressure in both directions. The mechanism is mapped — it activates the body's nitric oxide system to relax blood vessels and protect heart tissue. Zero of these findings have been tested in humans. The animal evidence is genuinely broader than most cardiac research peptides, but it's still rodent data, mostly from one research group. People using BPC-157 for heart concerns are running their own uncontrolled experiment.
BPC-157, a 15-amino-acid peptide derived from human gastric juice, has accumulated an extensive preclinical cardiovascular dataset over the past two decades. In rat and mouse models, it has reversed drug-induced arrhythmias, reduced myocardial infarction damage, counteracted congestive heart failure, lowered pulmonary hypertension, and produced endothelium-dependent vasodilation through a mapped nitric oxide signaling pathway.[1] The consistency of these findings across multiple research groups and cardiac injury models is unusual for a single peptide. The absence of any human cardiac data is equally striking. Every cardiovascular finding for BPC-157 comes from rodents. For a comprehensive overview of BPC-157 research across all organ systems, see BPC-157: The Body Protection Compound and What the Research Shows. For the broader story of how this peptide went from obscure gastric research to internet phenomenon, see The Real BPC-157 Story: 544 Papers, 30 Human Subjects, 50 Million Views.
Key Takeaways
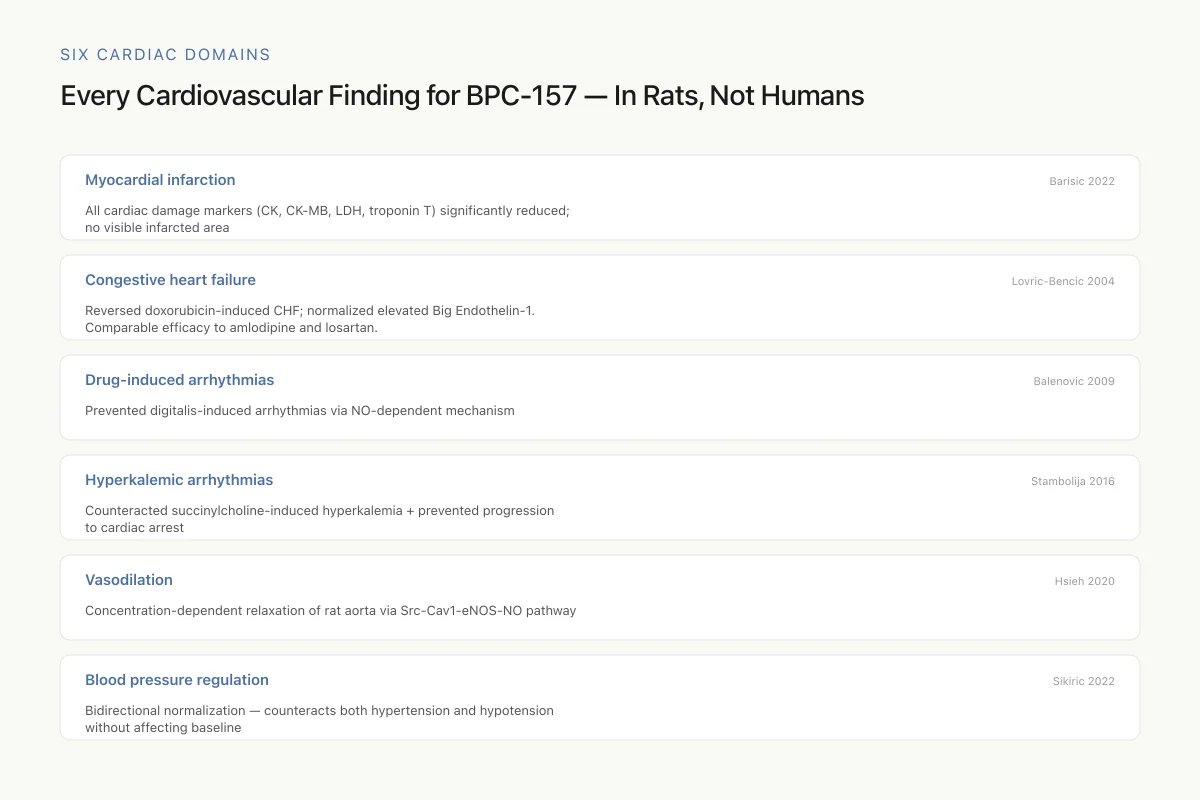
- BPC-157 at doses of 10 ng/kg and 10 micrograms/kg significantly reduced all cardiac necrosis markers (CK, CK-MB, LDH, troponin T) in isoprenaline-induced myocardial infarction in rats, with no visible infarcted area on gross examination (Barisic et al., Biomedicines, 2022)
- BPC-157 produces concentration-dependent vasodilation through a mapped Src-Caveolin-1-eNOS-NO pathway, confirmed by selective inhibitor experiments (Hsieh et al., Scientific Reports, 2020)
- The peptide inhibited methyldigoxin-induced arrhythmias through NO-system interaction, with L-NAME attenuating and L-arginine enhancing the antiarrhythmic effect (Balenovic et al., Regulatory Peptides, 2009)
- BPC-157 reversed doxorubicin-induced congestive heart failure and normalized elevated Big Endothelin-1 levels in rats, with efficacy comparable to amlodipine and losartan (Lovric-Bencic et al., 2004)
- BPC-157 counteracted succinylcholine-induced hyperkalemia and cardiac arrhythmias in rats, suggesting potential relevance to perioperative cardiac complications (Stambolija et al., European Journal of Pharmacology, 2016)
- A 2024 review extended these findings to vascular and multi-organ failure models, confirming BPC-157's cytoprotective effects across occlusion/reperfusion injury paradigms (Sikiric et al., Inflammopharmacology, 2024)
The Nitric Oxide Pathway: BPC-157's Cardiovascular Mechanism
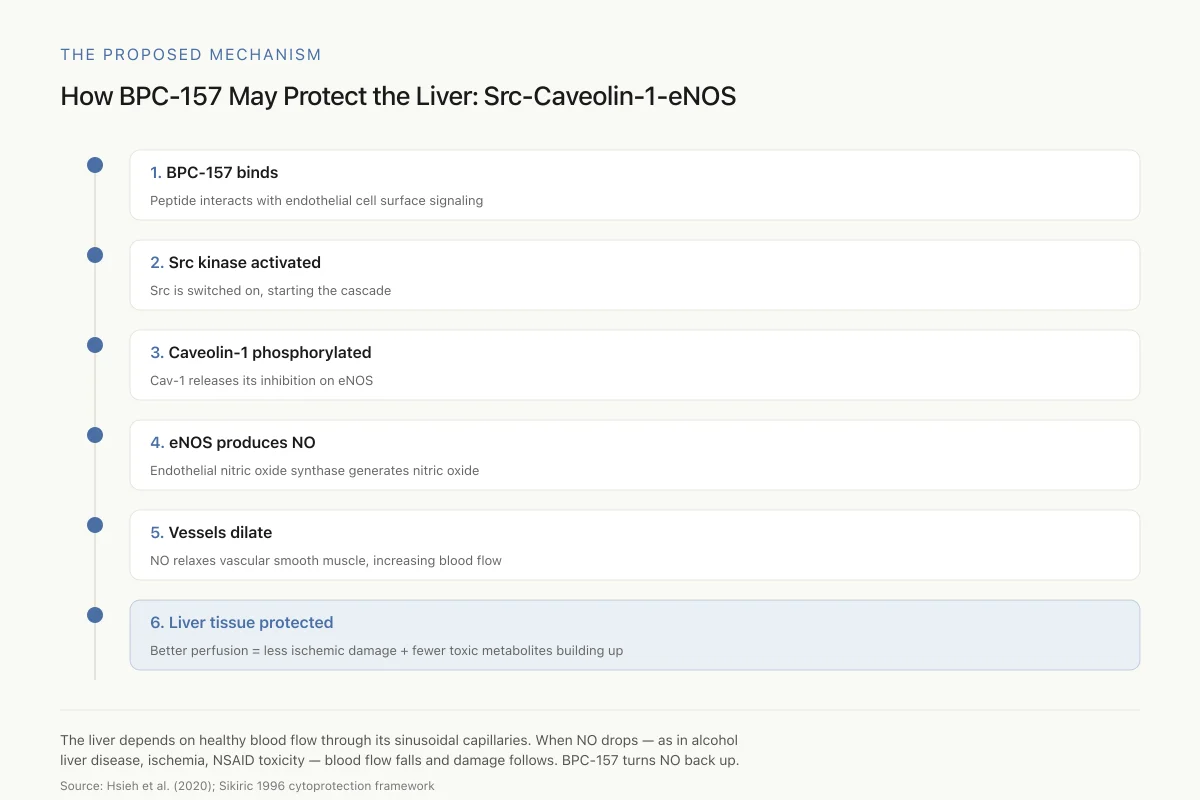
The most mechanistically detailed cardiovascular study of BPC-157 was published by Hsieh et al. in Scientific Reports in 2020.[2] Using isolated rat aorta preparations, the researchers mapped the vasodilation pathway step by step.
BPC-157 produced concentration-dependent relaxation of aortic rings. Removing the endothelium nearly eliminated the effect, confirming it is endothelium-dependent. Through a series of selective inhibitor experiments, the signaling cascade was identified:
- BPC-157 activates Src kinase (confirmed by increased Src phosphorylation)
- Activated Src phosphorylates Caveolin-1 (Cav-1)
- Phosphorylated Cav-1 releases its inhibitory binding to endothelial nitric oxide synthase (eNOS)
- Free eNOS produces nitric oxide (NO)
- NO activates soluble guanylyl cyclase, producing cGMP
- cGMP causes vascular smooth muscle relaxation
Each step was confirmed by blocking it with specific inhibitors: PP2 (Src inhibitor), L-NAME (eNOS inhibitor), and ODQ (guanylyl cyclase inhibitor) all attenuated BPC-157's vasodilatory effect. This level of pathway mapping is rare in BPC-157 research and provides the strongest mechanistic evidence for how the peptide affects the cardiovascular system.
The Proposed Mechanism
How BPC-157 May Protect the Liver: The Src-Caveolin-1-eNOS Pathway
A signaling cascade that ends in more nitric oxide, better hepatic blood flow, and reduced liver damage
The liver depends on healthy blood flow through its sinusoidal capillaries. When NO production drops — as it does in alcohol-related liver disease, ischemia, and NSAID toxicity — blood flow falls and damage follows. BPC-157 seems to turn the NO signal back up.
Source: Hsieh et al. (2020); Sikiric 1996 cytoprotection framework
 View as image
View as imageThe NO pathway also underlies BPC-157's antiarrhythmic effects. Balenovic et al. (2009) demonstrated that BPC-157 prevented methyldigoxin-induced arrhythmias in rats, and that L-NAME (an NOS inhibitor) attenuated this protection while L-arginine (the NO precursor) enhanced it.[3] This bidirectional modulation by NO-pathway components strongly implicates NO as the mediator of BPC-157's cardiac rhythm effects.
Myocardial Infarction: Preventing and Reversing Heart Damage
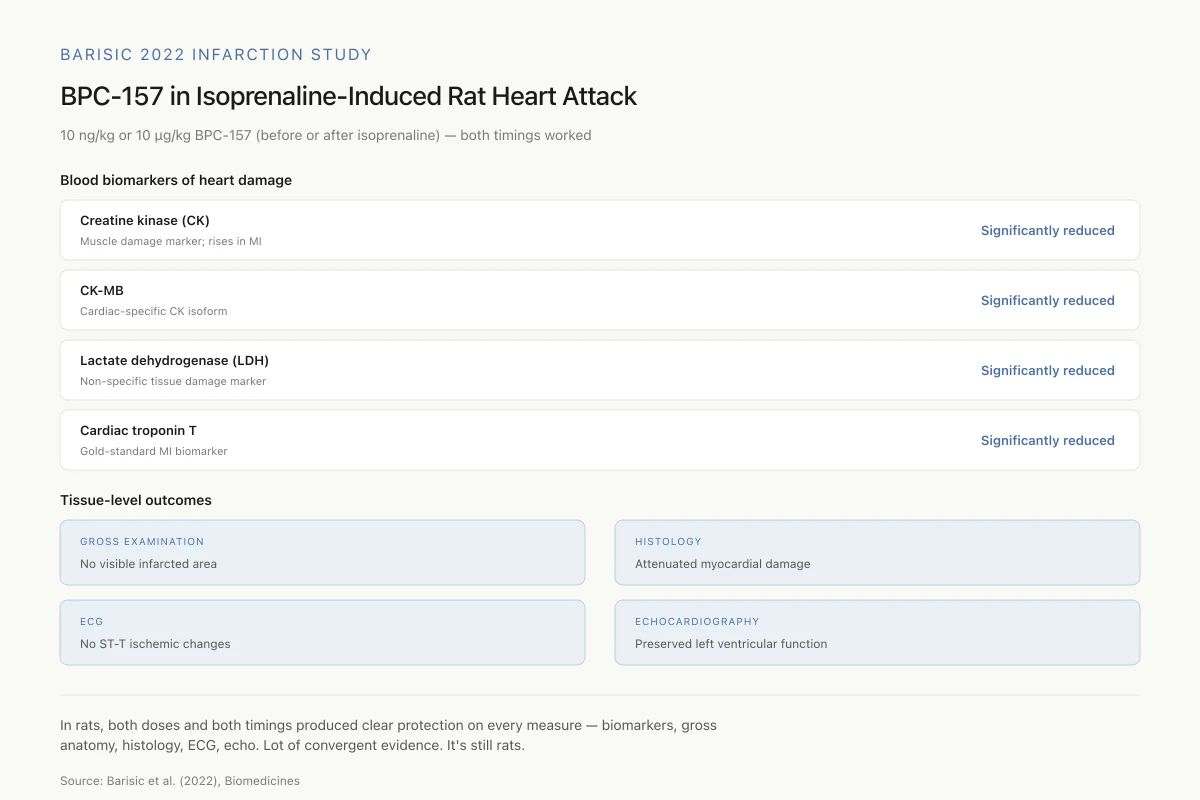
Barisic et al. (2022) tested BPC-157 in an isoprenaline-induced myocardial infarction model in rats.[4] Isoprenaline (isoproterenol) is a beta-adrenergic agonist that causes myocardial necrosis through excessive stimulation and oxygen demand, producing a pattern of damage that mimics aspects of human myocardial infarction.
BPC-157 was given at two doses (10 ng/kg and 10 micrograms/kg, intraperitoneally) either 30 minutes before or 5 minutes after isoprenaline administration. Both timing protocols and both doses produced significant results:
- All cardiac necrosis markers (creatine kinase, CK-MB, lactate dehydrogenase, cardiac troponin T) were significantly reduced
- Gross examination showed no visible infarcted area in treated rats
- Histological analysis confirmed attenuated myocardial damage
- ECG showed no ST-T ischemic changes (versus clear ischemic patterns in untreated rats)
- Echocardiography confirmed preservation of systolic left ventricular function
Barisic 2022 Infarction Study
BPC-157 in Isoprenaline-Induced Rat Heart Attack: The Numbers
10 ng/kg or 10 µg/kg BPC-157, given 30 min before or 5 min after isoprenaline — both timings and doses worked
In rats, both doses and both timing protocols produced clear protection on every measure — biomarkers, gross anatomy, histology, ECG, echocardiography. That's a lot of convergent evidence for the same effect. It's still rats.
Source: Barisic et al. (2022), Biomedicines
 View as image
View as imageThe study also showed that BPC-157 counteracted the early multi-organ failure that occurs within 30 minutes of isoprenaline-induced infarction, suggesting cardioprotection extends to systemic vascular effects. The finding that both pre-treatment and early post-treatment were effective is noteworthy: most cardioprotective agents are only tested as pre-treatments, but clinical relevance requires post-injury efficacy.
Congestive Heart Failure: Reversing Drug-Induced Cardiomyopathy
Lovric-Bencic et al. (2004) tested BPC-157 against doxorubicin-induced congestive heart failure in rats and mice.[5] Doxorubicin is an anthracycline chemotherapy drug with well-known cardiotoxicity: it causes dose-dependent cardiomyopathy that progresses to congestive heart failure, a major clinical problem in cancer treatment.
BPC-157 reversed the signs of doxorubicin-induced CHF and normalized elevated plasma concentrations of Big Endothelin-1, a vasoconstrictor peptide that increases in heart failure. The efficacy was comparable to amlodipine (a calcium channel blocker) and losartan (an angiotensin receptor blocker), both established cardiovascular drugs. This comparison to approved medications is significant because it places BPC-157's effect magnitude in a clinically interpretable context, even though the comparison was made in rodents rather than humans.
Big Endothelin-1 is elevated in human heart failure and correlates with disease severity and prognosis. The normalization of this biomarker by BPC-157 suggests the peptide may address the underlying vascular dysfunction of heart failure rather than merely alleviating symptoms. Endothelin-1 is a potent vasoconstrictor produced by damaged endothelium; its elevation in heart failure reflects endothelial dysfunction that worsens cardiac output through increased afterload. If BPC-157 restores endothelial function (consistent with its eNOS-activating mechanism), the reduction in Endothelin-1 would be a downstream consequence of endothelial repair rather than a direct pharmacological blockade. This mechanistic interpretation, if correct, positions BPC-157 as a restorative agent rather than a symptom manager, though the distinction requires human data to validate.
The doxorubicin cardiomyopathy model is also clinically relevant because chemotherapy-induced cardiotoxicity is a growing problem. As cancer survival rates improve, the long-term cardiac damage from anthracycline chemotherapy affects an increasing number of patients. Current cardioprotective strategies during chemotherapy are limited to dose reduction, dexrazoxane (the only approved cardioprotective agent), and standard heart failure medications after damage occurs. A peptide that could prevent or reverse anthracycline cardiotoxicity would address an unmet clinical need, but the distance from a rat model to clinical application remains vast.
Arrhythmias: Protection Across Multiple Models
Six Cardiac Domains
Every Cardiovascular Finding for BPC-157 — In Rats, Not Humans
The breadth of cardiac protection is genuinely unusual. The absence of human data is equally notable.
Six cardiovascular domains. One consistent theme: protection. One overwhelming limitation: exclusively rodent evidence. BPC-157 shows a breadth of cardiac effects unusual for a single peptide — but the translation to human cardiovascular disease has never been tested.
Source: Barisic 2022; Lovric-Bencic 2004; Balenovic 2009; Stambolija 2016; Hsieh 2020; Sikiric 2022
 View as image
View as imageBPC-157's antiarrhythmic effects have been demonstrated across several distinct arrhythmia models, each testing a different mechanism of cardiac rhythm disturbance.
Digitalis toxicity. Balenovic et al. (2009) showed BPC-157 prevented methyldigoxin-induced arrhythmias through NO-dependent mechanisms.[3] Digitalis toxicity is a clinical problem in patients taking digoxin for heart failure and atrial fibrillation, where narrow therapeutic windows make toxicity common.
Hyperkalemia. Stambolija et al. (2016) demonstrated that BPC-157 counteracted succinylcholine-induced hyperkalemia and the resulting cardiac arrhythmias in rats.[6] Hyperkalemic arrhythmias are a clinical emergency in surgical, burn, and crush injury settings. BPC-157 attenuated the rise in serum potassium and prevented the progression from arrhythmia to cardiac arrest in treated animals.
Ischemia-reperfusion. The comprehensive 2022 review by Sikiric et al. documented BPC-157's ability to reduce arrhythmia duration during hypoxia and reoxygenation in isolated guinea pig hearts, and to treat ventricular tachycardia, bradycardia, T-wave elevation, QTc prolongation, and asystole in various rat models.[1]
The breadth of arrhythmia models in which BPC-157 shows effects is unusual. Most antiarrhythmic agents are effective in one or two arrhythmia types; BPC-157 appears to have a more general stabilizing effect on cardiac rhythm, consistent with its mechanism acting through a fundamental pathway (NO/eNOS) rather than a specific ion channel.
This mechanistic distinction matters. Current antiarrhythmic drugs are classified by the Vaughan Williams system into four classes based on which ion channel they target: sodium channels (Class I), beta-adrenergic receptors (Class II), potassium channels (Class III), or calcium channels (Class IV). Each class works for specific arrhythmia types and can paradoxically cause other arrhythmias (proarrhythmic effects). BPC-157 does not fit this classification. Its action through NO signaling operates upstream of individual ion channels, potentially stabilizing the electrical environment of the heart more broadly.
The clinical implication, if confirmed in humans, would be significant: a compound that protects against multiple arrhythmia types without the proarrhythmic risks of current antiarrhythmic drugs would address an unmet need in cardiology. The caveat remains that this potential is entirely theoretical for human hearts. The guinea pig isolated heart preparations and rat models used in these studies have different ion channel compositions and conduction properties than the human heart.
Blood Pressure: The Bidirectional Effect
A Regulator, Not a Driver
BPC-157's Unusual Bidirectional Blood Pressure Effect
Unlike conventional BP drugs that push in one direction, BPC-157 normalizes toward baseline — in rats
Amlodipine lowers BP whether it's 180 or 120. Norepinephrine raises it regardless of starting point. Most cardiovascular drugs are one-way. BPC-157 appears to read the state of the system and push it back toward normal — a genuinely unusual property, if it translates to humans (which is untested).
Source: Sikiric et al. (2022), Biomedicines
 View as image
View as imageOne of BPC-157's most distinctive properties is its bidirectional effect on blood pressure. Rather than simply lowering or raising blood pressure, BPC-157 appears to normalize it toward baseline from either direction. In studies using L-NAME (which blocks NO production and causes hypertension), BPC-157 counteracted the blood pressure elevation. In studies using L-arginine (which increases NO and can cause hypotension), BPC-157 counteracted the blood pressure drop.[1] Basal blood pressure in normal animals was not affected.
This normalization pattern distinguishes BPC-157 from conventional antihypertensive or vasopressor drugs, which push blood pressure in one direction regardless of the starting point. Amlodipine lowers blood pressure whether it starts at 180 mmHg or 120 mmHg. Norepinephrine raises it regardless of starting point. BPC-157 appears to act as a regulator rather than a driver, bringing the system back toward its set point.
The mechanistic explanation likely involves the NO pathway identified by Hsieh et al. Nitric oxide is itself a bidirectional regulator of vascular tone: it is produced in response to both shear stress (which increases in hypertension) and endothelial dysfunction (which occurs in both hyper- and hypotensive states). If BPC-157 enhances eNOS function rather than simply increasing NO production, it could restore the endothelium's intrinsic ability to regulate vascular tone, explaining the bidirectional effect.
This interpretation remains speculative because the blood pressure normalization data comes from whole-animal studies where multiple compensatory systems are active. Isolated vessel preparations (like the aortic rings used by Hsieh et al.) show only vasodilation, not bidirectional effects, suggesting the normalization may require intact systemic regulatory circuits rather than being an intrinsic property of BPC-157's molecular action.
Vascular Protection and Multi-Organ Failure
Sikiric et al. (2024) expanded the cardiovascular evidence with new studies on BPC-157 in occlusion and occlusion-like syndromes.[7] Vascular occlusion causes ischemia followed by reperfusion injury, a central mechanism in heart attacks, strokes, organ transplantation, and surgical complications. BPC-157 demonstrated cytoprotective effects across multiple organ systems during these occlusion-reperfusion challenges, consistent with its documented NO-pathway mechanism.
Staresinic et al. (2022) reviewed BPC-157's effects on striated, smooth, and cardiac muscle, providing a comprehensive assessment of how the peptide interacts with all three muscle types relevant to cardiovascular function.[8] The review documented effects on both vascular smooth muscle tone and cardiac contractility, connecting the vasodilation evidence from Hsieh et al. to the cardiac protection evidence from the infarction and heart failure studies. The convergence of effects across all three muscle types suggests BPC-157 acts on fundamental cellular protective mechanisms rather than tissue-specific pathways.
The vascular protection findings are particularly relevant in the context of angiogenesis. BPC-157 has been documented to increase vascular endothelial growth factor receptor 2 (VEGFR2) expression, promoting the formation of new blood vessels. In the cardiovascular context, enhanced angiogenesis could contribute to collateral vessel formation after myocardial infarction, improving blood supply to damaged heart tissue. This pro-angiogenic effect has been demonstrated in wound healing models and may partially explain why BPC-157 preserves heart function after ischemic injury: by rapidly establishing alternative blood supply routes to ischemic myocardium.
The connection between BPC-157's gastroprotective origins and its cardiovascular effects is not coincidental. The gastrointestinal tract is one of the most vascularized organ systems in the body, and gastric protection inherently involves vascular protection. The endothelium that lines gastric blood vessels uses the same NO/eNOS machinery that lines coronary arteries. BPC-157's original characterization as a gastroprotective agent may have been observing the same endothelial protective mechanism that produces cardiovascular effects when studied directly.
For other peptide approaches to cardiac repair including thymosin beta-4 and GHK-Cu, see Thymosin Beta-4 and Cardiac Repair: Healing Heart Muscle, GHK-Cu and Cardiac Tissue: The Copper Peptide's Heart Research, and Peptide Approaches to Heart Muscle Regeneration After Heart Attack.
The Critical Limitation: No Human Data
Safety
ModerateNo human cardiac safety data + drug interaction risk
Concern
Rodent studies show no adverse cardiac effects in healthy rats, but patients with heart disease have compromised cardiac reserve and are usually on multiple medications. A vasodilatory peptide that normalizes BP in healthy rats could cause dangerous hypotension in a patient already on ACE inhibitors, beta-blockers, and diuretics. Drug-drug interactions with standard cardiac medications have never been studied.
What the research says
If you have heart disease, are on cardiac medications, or have a cardiovascular history, BPC-157 is not a safe experiment. The animal evidence doesn't cover what happens when you combine a vasodilatory peptide with three or four other vasodilatory drugs.
Particularly relevant for: Anyone with cardiovascular disease, on BP meds, or with arrhythmia history
What to do
If you still want to try BPC-157, discuss with a cardiologist first. Keep baseline and follow-up blood pressure readings. Monitor for symptoms of hypotension (dizziness, fatigue, syncope).
Current literature; clinical pharmacology principles
Every finding described above comes from rats, mice, or isolated animal tissue preparations. The cardiovascular evidence for BPC-157 is exclusively preclinical. No human cardiac safety trial, no human efficacy study, no pharmacokinetic analysis of BPC-157 in human cardiovascular tissue, and no human dosing study for cardiac indications have been published.
This is not a minor caveat. It is the defining feature of the BPC-157 cardiovascular evidence base. The history of drug development is filled with compounds that showed dramatic cardioprotection in rodents but failed or caused harm in humans. Rodent hearts differ from human hearts in size, heart rate (rodents: 300-600 bpm; humans: 60-100 bpm), coronary anatomy, ion channel expression, and susceptibility to arrhythmias. Effects observed in rat infarction models frequently do not predict human outcomes.
Additionally, the vast majority of BPC-157 cardiovascular studies come from a single research group led by Predrag Sikiric at the University of Zagreb. While their work is prolific and internally consistent, independent replication by other laboratories is limited. This is a common concern across BPC-157 research, not specific to cardiovascular studies. Independent replication is the standard by which preclinical findings graduate from "interesting" to "reliable."
The doses used in animal studies (10 ng/kg to 10 micrograms/kg) cannot be directly extrapolated to human dosing without pharmacokinetic data. How BPC-157 distributes to cardiac tissue in humans, what its half-life is in the human cardiovascular system, and whether the NO-pathway activation observed in rat aorta occurs in human endothelium are all unknown.
People who use gray-market BPC-157 and report cardiovascular effects are providing anecdotes, not evidence. Self-reported improvements in blood pressure, exercise tolerance, or heart rate variability cannot be attributed to BPC-157 without controlled conditions, objective measurements, and comparison groups.
What a Human Cardiac Trial Would Need
If BPC-157 were to enter human cardiovascular development, several questions would need to be addressed in sequence.
Pharmacokinetics. How does BPC-157 distribute to cardiac tissue after injection in humans? What is its half-life in the cardiovascular system? Does it cross the blood-heart barrier in sufficient concentrations to replicate the tissue-level effects seen in rodents? The doses that protected rat hearts (10 ng/kg to 10 micrograms/kg) span a 1,000-fold range; human dose selection would require pharmacokinetic data to identify the therapeutic window.
Safety in cardiac patients. The animal studies show no adverse cardiac effects in healthy animals, but patients with heart disease have compromised cardiac reserve. A vasodilatory peptide that normalizes blood pressure in healthy rats might cause problematic hypotension in a patient already on ACE inhibitors, beta-blockers, and diuretics. Drug-drug interactions with standard cardiac medications have not been studied.
Endpoint selection. Cardiac clinical trials require hard endpoints: mortality, hospitalization, major adverse cardiac events. The animal studies used biomarker and imaging surrogates (troponin levels, echocardiographic function, ECG changes) that are relevant but insufficient for human drug approval. A trial designed to show mortality benefit in heart failure or infarction would require thousands of patients and years of follow-up.
Route of administration. The animal studies used intraperitoneal injection, which is not used clinically in humans. Subcutaneous, intravenous, or oral routes would each produce different pharmacokinetic profiles. The oral route is attractive (BPC-157 was originally studied as a gastric peptide), but oral bioavailability for cardiac indications has not been established.
Regulatory pathway. BPC-157 is not patentable as a naturally derived peptide sequence, which limits the commercial incentive for pharmaceutical companies to invest in the expensive clinical trials required for cardiac drug approval. This economic reality, not scientific skepticism, is likely the primary reason no human cardiac trial has been conducted. The animal evidence is strong enough to justify clinical investigation; the business case may not be.
The gap between preclinical promise and clinical reality is a familiar story in cardiovascular drug development. Many compounds that protected rodent hearts failed in human trials, and many that failed in early human trials were later found to work at different doses or in different patient populations. BPC-157's cardiovascular evidence base is more extensive and more mechanistically coherent than most preclinical cardiovascular candidates, but it remains exactly that: preclinical. The strength of the animal data makes the absence of human trials notable. The weakness of the evidence base, its concentration in a single research group and its exclusive reliance on rodent models, makes the absence of independent replication and human testing equally notable. Both observations are true simultaneously, and both should inform how this evidence is interpreted.
The cardiovascular peptide research landscape extends beyond BPC-157. Thymosin beta-4 has been studied for cardiac regeneration after myocardial infarction, with evidence for promoting cardiomyocyte survival and neovascularization. GHK-Cu (copper peptide) has shown effects on cardiac fibrosis and tissue remodeling. Natriuretic peptides (ANP, BNP) are established biomarkers and therapeutic targets in heart failure. BPC-157's unique contribution to this landscape is its breadth: no other single peptide has demonstrated effects across arrhythmias, infarction, heart failure, pulmonary hypertension, and vascular tone in animal models. Whether this breadth reflects a genuinely versatile cardiovascular peptide or an artifact of how the studies were designed and reported is a question that only independent replication and human trials can answer.
The Bottom Line
BPC-157 has demonstrated a coherent pattern of cardiovascular protection in animal models: vasodilation through a mapped NO pathway, myocardial infarction damage reduction, congestive heart failure reversal, antiarrhythmic effects across multiple models, and bidirectional blood pressure normalization. The mechanistic evidence is stronger than for most peptides in preclinical development. The clinical evidence is nonexistent. No human cardiovascular trial has been conducted or, as of 2026, publicly registered. The animal data suggests a peptide with genuine cardiovascular pharmacology worth investigating in human studies. It does not support using BPC-157 for cardiovascular self-treatment. The distance between "works in rats" and "works in humans" is measured in clinical trials that have not occurred.
Sources & References
- 1RPEP-06502·Sikiric, Predrag et al. (2022). “Stable Gastric Pentadecapeptide BPC 157 as Useful Cytoprotective Peptide Therapy in the Heart Disturbances, Myocardial Infarction, Heart Failure, Pulmonary Hypertension, Arrhythmias, and Thrombosis Presentation..” Biomedicines.Study breakdown →PubMed →↩
- 2RPEP-04861·Hsieh, Ming-Jer et al. (2020). “BPC-157 Relaxes Blood Vessels Through a Mapped Molecular Pathway Involving Nitric Oxide.” Scientific reports.Study breakdown →PubMed →↩
- 3RPEP-01443·Unknown (2009). “Balenovic 2009 Inhibition Of Methyldigoxininduced Arrhythmiasthmias.” .Study breakdown →↩
- 4RPEP-06001·Barisic, Ivan et al. (2022). “BPC-157 Protects Against Heart Attack Damage and Multi-Organ Failure in Rats.” Biomedicines.Study breakdown →PubMed →↩
- 5RPEP-00944·Lovric-Bencic, Martina et al. (2004). “BPC-157 Reverses Doxorubicin-Induced Heart Failure and Big Endothelin-1 Elevation in Rats.” Journal of pharmacological sciences.Study breakdown →PubMed →↩
- 6RPEP-03120·Stambolija, Vasilije et al. (2016). “BPC 157 Peptide Completely Eliminated Dangerous Heart Rhythm Problems and High Potassium Caused by Succinylcholine in Rats.” European journal of pharmacology.Study breakdown →PubMed →↩
- 7RPEP-09268·Sikiric, Predrag et al. (2024). “BPC 157: A Gastric Peptide That May Protect Multiple Organs by Rescuing Blood Flow During Vascular Crises.” Inflammopharmacology.Study breakdown →PubMed →↩
- 8RPEP-06515·Staresinic, Mario et al. (2022). “Stable Gastric Pentadecapeptide BPC 157 and Striated, Smooth, and Heart Muscle..” Biomedicines.Study breakdown →PubMed →↩