Argireline: The 'Botox in a Bottle' Peptide
Cosmetic Peptides
48.9% Efficacy
In a randomized, placebo-controlled trial in 60 Chinese subjects, argireline cream produced 48.9% total anti-wrinkle efficacy versus 0% for placebo, with significant reductions in all roughness parameters.
Wang et al., Am J Clin Dermatol, 2013
Wang et al., Am J Clin Dermatol, 2013
If you only read one thing
Argireline is a tiny peptide in anti-wrinkle creams that tries to relax your facial muscles — the same basic idea as Botox, but applied as a cream instead of an injection. Clinical studies show it does reduce wrinkle depth by about 17–30%, which is real but modest. The catch: less than 0.01% of what you put on your skin actually gets through to where it needs to work. It's extremely safe and does something, but calling it 'Botox in a bottle' is a stretch.
In 2002, a team at the Universidad Miguel Hernandez in Alicante, Spain, published the first study on a synthetic hexapeptide designed to reduce wrinkles by interfering with the molecular machinery of neurotransmitter release.[1] The peptide, Ac-EEMQRR-NH2, was modeled on the N-terminal end of SNAP-25, one of three proteins that form the SNARE complex required for acetylcholine release at the neuromuscular junction. Botulinum toxin (Botox) paralyzes muscles by cleaving SNAP-25. The peptide was designed to compete with SNAP-25 for binding partners, destabilizing the SNARE complex without destroying the protein. Applied topically in an oil/water emulsion at 10% concentration, it reduced wrinkle depth by up to 30% after 30 days. The developers named it Argireline.
Two decades later, argireline (now officially acetyl hexapeptide-8) is one of the most commercially successful cosmetic peptides in the world. It appears in thousands of skincare products marketed as a topical alternative to botulinum toxin injections. The marketing claim, "Botox in a bottle," has driven enormous consumer interest. The scientific reality is more complicated: argireline does appear to reduce wrinkle depth in controlled studies, but the magnitude of effect is far smaller than injectable botulinum toxin, and the fundamental challenge of getting a 889-Da hydrophilic peptide through the skin's lipophilic barrier remains largely unsolved.
This article examines the mechanism, the clinical evidence, the penetration problem, and where argireline fits in the broader landscape of cosmetic peptides. For how argireline compares to other neurotransmitter-inhibiting peptides, see Neurotransmitter-Inhibiting Peptides: How They Relax Facial Muscles. For the full taxonomy of cosmetic peptide types, see Every Type of Cosmetic Peptide: Signal, Carrier, Neurotransmitter, and Enzyme Inhibitor.
Key Takeaways
- "Botox in a bottle" is marketing — no study has ever compared argireline head-to-head with real Botox.
- Less than 0.01% of the cream actually makes it through your skin to where it could work.
- In controlled trials, wrinkle depth dropped by roughly 17–30% after a month of twice-daily use.
- Some of that improvement may just come from the moisturizer in the cream, not the peptide itself.
- Argireline is extraordinarily safe — over 100 million times less toxic than real Botox.
- Argireline works by crowding the muscle-signaling system, not destroying it the way Botox does.
- The strongest trial showed 48.9% improvement over placebo, but that measures surface texture, not visible muscle relaxation.
How Argireline Works: The SNARE Complex
To understand argireline, you need to understand how muscles receive the signal to contract.
When a motor neuron fires, an electrical impulse travels down the axon to the nerve terminal. At the terminal, the impulse triggers calcium influx, which activates the SNARE (soluble N-ethylmaleimide-sensitive factor attachment protein receptor) complex. This complex consists of three proteins: syntaxin-1 and SNAP-25 on the presynaptic membrane, and VAMP (vesicle-associated membrane protein, also called synaptobrevin) on the synaptic vesicle. When these three proteins assemble, they pull the vesicle membrane and the cell membrane together, forcing them to fuse. This fusion releases acetylcholine into the synaptic cleft, where it binds receptors on the muscle fiber and triggers contraction.
Botulinum toxin type A, the active ingredient in Botox, Dysport, and Xeomin, works by enzymatically cleaving SNAP-25. Once SNAP-25 is cleaved, the SNARE complex cannot assemble, vesicle fusion stops, acetylcholine release is blocked, and the muscle is paralyzed. The effect lasts 3-6 months because the neuron must synthesize new SNAP-25 protein to restore function.
Argireline takes a different approach. Instead of destroying SNAP-25, it competes with it. The hexapeptide Ac-EEMQRR-NH2 mimics the N-terminal domain of SNAP-25 and competes for binding to VAMP and syntaxin-1.[1] By occupying binding sites on these partner proteins, argireline reduces the efficiency of SNARE complex assembly. Fewer functional SNARE complexes means less vesicle fusion, less acetylcholine release, and weaker muscle contraction. The effect is competitive inhibition: argireline reduces neurotransmitter release but does not eliminate it. This is fundamentally different from botulinum toxin's irreversible proteolytic cleavage.
Blanes-Mira et al. demonstrated that argireline inhibited neurotransmitter release with a potency similar to botulinum toxin type A in cell-based assays, but with much lower efficacy (maximum inhibition).[1] In plain terms: argireline reduces the signal, but it cannot shut it off entirely the way Botox does. This explains why clinical effects are measurable but modest compared to injectable neurotoxin.
Clinical Evidence
The Original Study (2002)
Blanes-Mira et al. tested a 10% argireline oil/water emulsion on healthy female volunteers, applied twice daily to the periorbital area.[1] Skin topography analysis showed wrinkle depth reduction of up to 17% after 15 days and up to 30% after 30 days. The study was small and open-label (not placebo-controlled), which limits the strength of these findings, but it established proof of concept.
Randomized Controlled Trial (2013)
The strongest clinical evidence comes from Wang et al., who conducted a randomized, placebo-controlled trial in 60 Chinese subjects (45 argireline, 15 placebo, 3:1 randomization).[2] Participants applied argireline cream to the periorbital area for 4 weeks. The total anti-wrinkle efficacy was 48.9% in the argireline group versus 0% in the placebo group. All roughness parameters (Ra, Rz, Rp, Rv) were significantly decreased in the argireline group (p < 0.01), while no significant changes occurred in the placebo group. This remains the highest-quality clinical trial on argireline, and the results, while positive, should be interpreted in context: 48.9% efficacy on a roughness scale is not the same as a 48.9% reduction in visible wrinkles. The measurement captured surface texture changes, not the dramatic smoothing that injectable botulinum toxin produces.
Head-to-Head Comparison with Matrixyl (2023)
A double-blind, randomized trial compared acetylhexapeptide-3 cream and palmitoyl pentapeptide-4 (Matrixyl) cream for crow's feet in 21 Indonesian women over 8 weeks.[3] Both peptide creams showed improvements in skin hydration, elasticity, and wrinkle grading compared to placebo, but neither produced dramatic results. The study's small sample size (7 per group) limits statistical power. For a deeper comparison of argireline with Matrixyl and other signal peptides, see Matrixyl (Palmitoyl Pentapeptide-4): The Anti-Wrinkle Peptide in Your Serum.
What the Trials Do Not Show
No published clinical trial has demonstrated that topical argireline produces effects comparable to injectable botulinum toxin. The "Botox in a bottle" marketing claim is not supported by head-to-head comparison data. The wrinkle reduction measured in clinical studies (17-30% depth reduction by surface profilometry) is real but modest. No study has shown that argireline produces visible muscle relaxation in facial muscles the way botulinum toxin does. The clinical effects may be partly due to moisturizing properties of the emulsion vehicle rather than the peptide's neuromuscular mechanism.
The Penetration Problem
The fundamental challenge for argireline is skin penetration. For the peptide to inhibit the SNARE complex at the neuromuscular junction, it must pass through the stratum corneum (the skin's outer barrier), traverse the epidermis and dermis, and reach the nerve terminals at the dermal-epidermal junction or deeper.
Argireline has a molecular weight of 889 Da and a partition coefficient (LogP) of -6.3. Both values work against skin penetration. The "rule of 500" in dermal pharmacology states that molecules above 500 Da penetrate the stratum corneum poorly. Argireline is nearly twice that threshold. Its strongly negative LogP indicates extreme hydrophilicity, meaning it partitions into water rather than the lipid-rich environment of the stratum corneum.
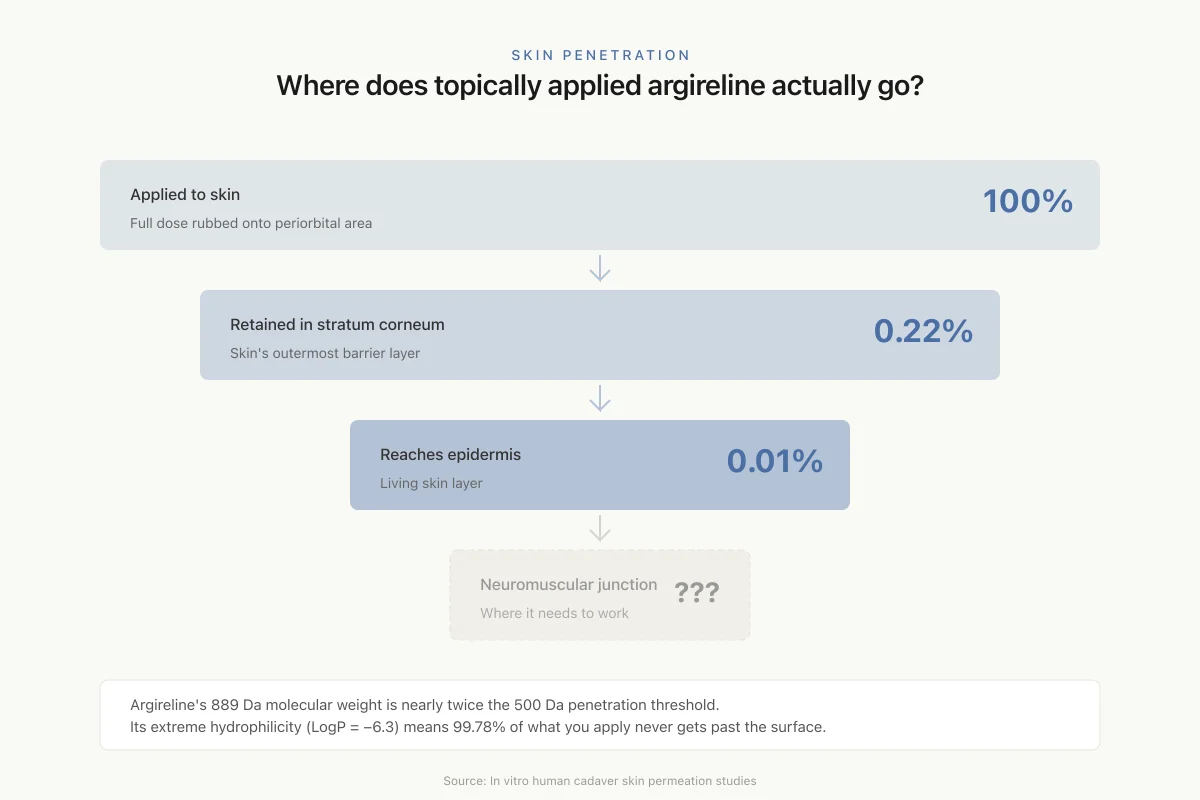
In vitro skin permeation studies using human cadaver skin quantified the problem. The vast majority of applied argireline remained on the skin surface and was washed away. Only 0.22% of the total dose was retained within the stratum corneum. Just 0.01% reached the epidermis. These numbers raise a fundamental question: can 0.01% of the applied dose reach the neuromuscular junction at sufficient concentration to compete with endogenous SNAP-25?
Skin Penetration
Where does topically applied argireline actually go?
Argireline's 889 Da molecular weight is nearly twice the 500 Da penetration threshold. Its extreme hydrophilicity (LogP = −6.3) means it prefers water over the lipid-rich skin barrier. Result: 99.78% of what you apply never gets past the surface.
Source: In vitro human cadaver skin permeation studies
 View as image
View as imageLim et al. addressed this challenge by chemically modifying argireline to improve skin permeation.[4] By reducing the zwitterionic character of the peptide through chemical modifications, they achieved approximately 30-fold increased permeation using iontophoresis (an electrical current that drives charged molecules through skin). Other approaches under investigation include microneedle delivery, liposomal encapsulation, and penetration-enhancing carrier peptides.
The penetration problem does not mean argireline is inactive when applied topically. The clinical trials show measurable wrinkle reduction. But the mechanism producing those results may not be the SNARE complex inhibition demonstrated in cell-based assays. Surface moisturization, mild anti-inflammatory effects, or local action on epidermal cells (which do not require deep penetration) could contribute to the observed clinical effects.
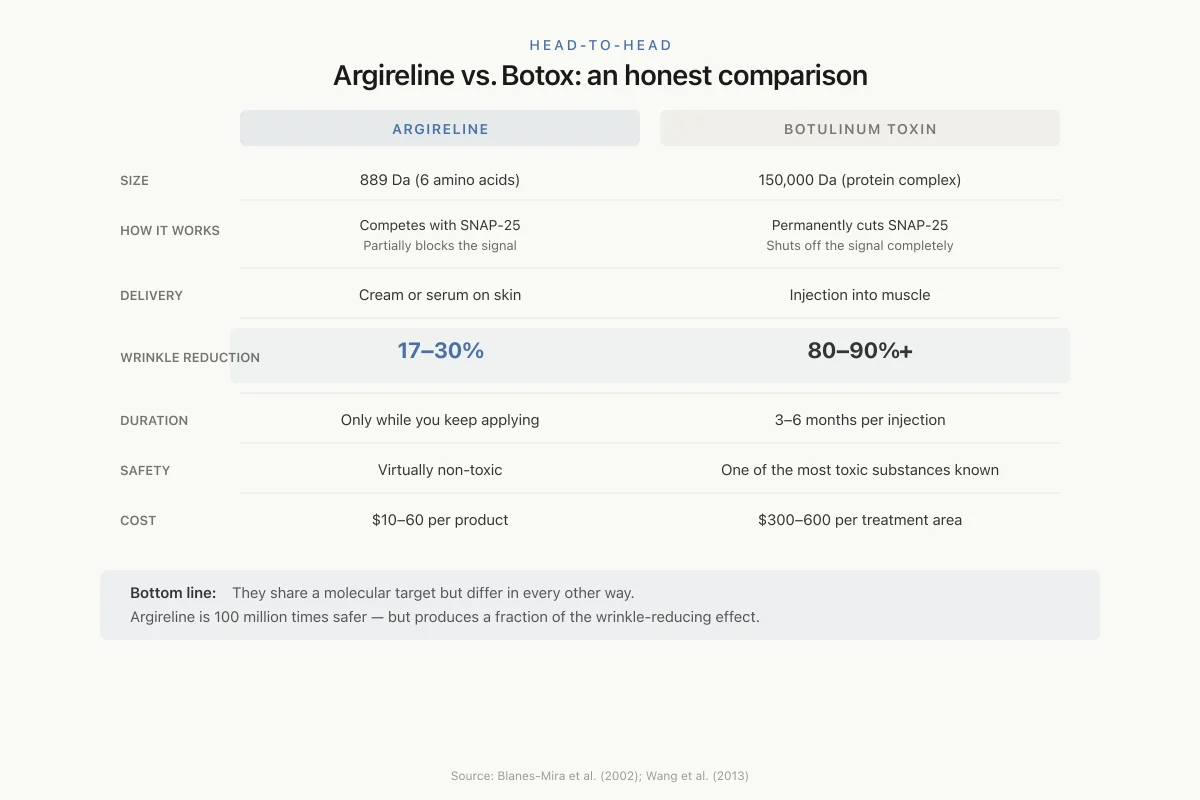
Argireline vs. Botulinum Toxin: An Honest Comparison
The comparison is commercially convenient but scientifically misleading. The two compounds share a molecular target (the SNARE complex) but differ in every other respect.
Head-to-Head
Argireline vs. Botox: an honest comparison
Argireline
Botulinum Toxin
Size
How it works
Delivery
Wrinkle reduction
How long it lasts
Safety margin
Cost
Bottom line: They share a molecular target (the SNARE complex) but differ in every other way. Argireline is 100 million times safer — but produces a fraction of the wrinkle-reducing effect.
Source: Blanes-Mira et al. (2002); Wang et al. (2013)
 View as image
View as imageThe safety margin is worth emphasizing. Argireline's LD50 exceeds 2,000 mg/kg, while botulinum toxin's LD50 is approximately 20 ng/kg. This represents a safety differential of over 100 million-fold. Argireline is, for practical purposes, non-toxic. Botulinum toxin is one of the most poisonous substances known, which is why it requires precise dosing by trained practitioners.
SNAP-8 and Next-Generation Variants
SNAP-8 (acetyl octapeptide-3) extends the argireline concept with an 8-amino-acid sequence (Ac-EEMQRRAD-NH2) rather than 6. The additional two residues (alanine and aspartic acid) were designed to improve binding specificity for the SNARE complex. SNAP-8 targets the same mechanism but may achieve more efficient SNARE complex destabilization because its longer sequence provides additional contact points with VAMP and syntaxin-1.
Leuphasyl (pentapeptide-18) takes a different approach entirely. Instead of targeting the SNARE complex directly, it mimics enkephalin and acts on opioid receptors to reduce catecholamine release, which indirectly reduces muscle contraction. The combination of argireline with leuphasyl has been marketed as producing synergistic effects, though independent clinical validation of this claim is limited.
The broader category of neurotransmitter-inhibiting cosmetic peptides continues to expand. For a comprehensive overview of how these peptides compare, see Neurotransmitter-Inhibiting Peptides: How They Relax Facial Muscles.
The Cosmetic Peptide Landscape
Argireline belongs to one of four functional categories of cosmetic peptides.
Neurotransmitter-inhibiting peptides (argireline, SNAP-8, leuphasyl) reduce muscle contraction signals. They target expression lines, the wrinkles caused by repeated facial movements.
Signal peptides (Matrixyl/palmitoyl pentapeptide-4, GHK-Cu) stimulate fibroblasts to produce collagen, elastin, and other extracellular matrix proteins. They target fine lines and overall skin texture. GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is the most extensively studied signal peptide, with evidence spanning wound healing, collagen synthesis, and gene expression modulation in skin regeneration.[5]
Carrier peptides deliver trace elements (particularly copper) to skin cells. GHK-Cu doubles as both a signal and carrier peptide.
Enzyme-inhibiting peptides block enzymes that degrade the extracellular matrix, such as matrix metalloproteinases (MMPs) that break down collagen and elastin.
These categories are not mutually exclusive. Some products combine peptides from multiple categories (e.g., argireline for expression lines plus Matrixyl for collagen stimulation) based on the rationale that targeting different aspects of skin aging simultaneously should produce better results than any single peptide. This combinatorial logic is plausible but largely untested in rigorous clinical studies. For the evidence on whether peptide serums deliver their promises, see Do Peptide Serums Actually Work? What Clinical Studies Show.
Skin Peptide Delivery: The Shared Challenge
The penetration problem is not unique to argireline. Nearly all cosmetic peptides face the same barrier. Collagen-stimulating peptides like Matrixyl must reach dermal fibroblasts, which sit beneath the epidermis. GHK-Cu must deliver copper ions to cells in the dermis. The stratum corneum's lipophilic barrier blocks hydrophilic peptides regardless of their mechanism.
Collagen hydrolysates represent an alternative delivery route. Oral collagen peptides bypass the skin barrier entirely, reaching dermal fibroblasts through the bloodstream after intestinal absorption. Aguirre-Cruz et al. reviewed evidence for both oral and topical collagen peptide delivery for skin protection.[6] Chae et al. demonstrated that fish-derived collagen peptides containing 3% Gly-Pro-Hyp tripeptide could prevent cortisol-induced collagen type I loss in human dermal fibroblasts by blocking glucocorticoid receptor signaling.[7]
The contrast is instructive: oral collagen peptides have clearer bioavailability evidence than topical argireline, even though both are marketed as anti-aging peptide products. The cosmetic peptide field broadly suffers from a disconnect between in vitro mechanism studies and in vivo delivery reality.
Formulation and Concentration
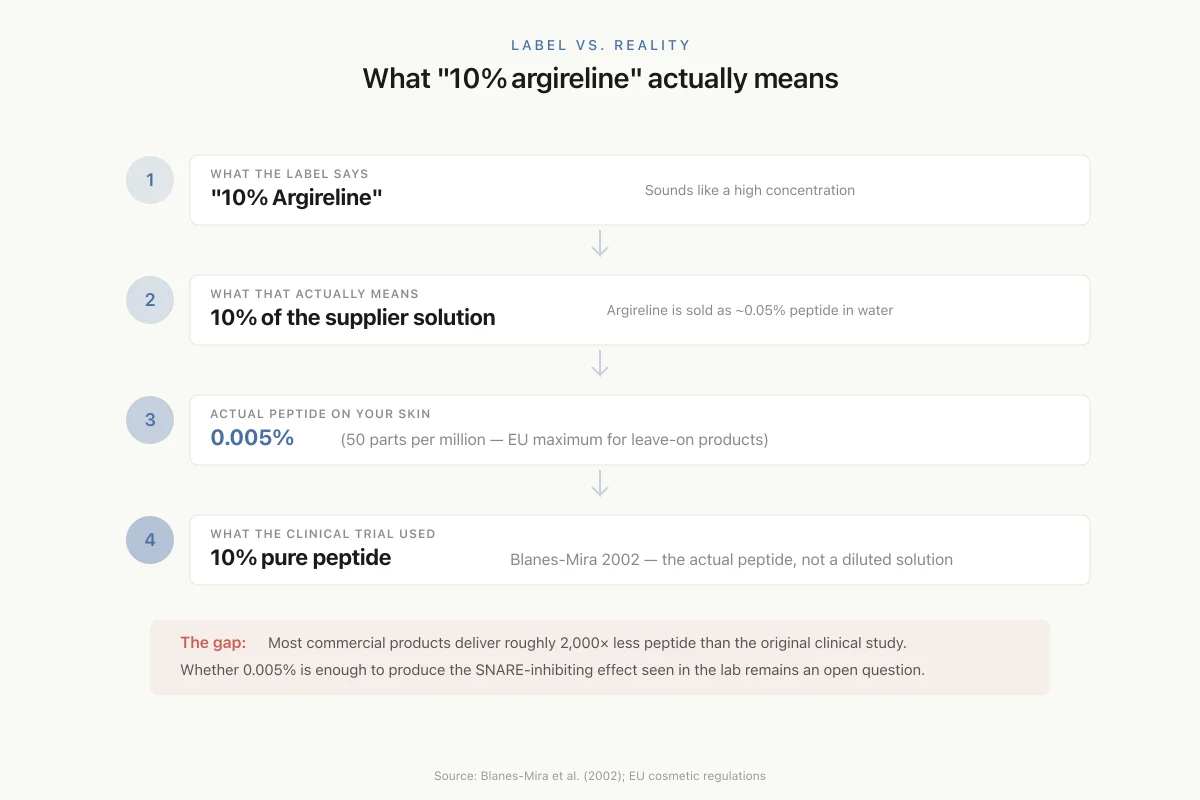
The concentration of argireline in commercial products varies widely, and label claims can be misleading. The original Blanes-Mira study used a 10% solution of the argireline peptide in an oil/water emulsion. However, argireline is commercially supplied as a solution (typically 0.05% peptide in water with preservatives), not as a pure peptide powder. When a product claims "10% argireline," it usually means 10% of the supplier's solution, which translates to approximately 0.005% (50 ppm) of the actual peptide. This is the concentration recommended by the EU for leave-on cosmetic products.
The distinction matters for evaluating efficacy claims. A product containing 0.005% active peptide is delivering a very different dose than one containing 10% pure peptide. The clinical trials used concentrations at the higher end, and it remains unclear whether the concentrations in most commercial products are sufficient to produce the measured clinical effects.
Label vs. Reality
What “10% argireline” actually means
What the label says
"10% Argireline"
Sounds like a high concentration
What that actually means
10% of the supplier solution
Argireline is sold as ~0.05% peptide dissolved in water + preservatives
Actual peptide on your skin
0.005%
That's 50 parts per million — the EU maximum for leave-on products
What the clinical trial used
10% pure peptide
Blanes-Mira 2002 used 10% of the actual peptide, not 10% of a diluted solution
The gap: Most commercial products deliver roughly 2,000× less peptide than the original clinical study used. Whether 0.005% is enough to produce the SNARE-inhibiting effect seen in the lab remains an open question.
Source: Blanes-Mira et al. (2002); EU cosmetic regulations
 View as image
View as imageVehicle formulation also affects delivery. The oil/water emulsion used in the original study was specifically designed to maximize contact time and hydration of the stratum corneum. Different formulation vehicles (gels, serums, creams, lotions) will produce different skin surface residence times, different hydration effects, and potentially different degrees of peptide penetration. A peptide serum in a hyaluronic acid base, for example, may improve skin hydration independently of argireline's neuromuscular mechanism, making it difficult to attribute wrinkle improvement to the peptide alone.
Safety and Regulatory Status
Argireline has an excellent safety profile. Acute toxicity testing shows LD50 values exceeding 2,000 mg/kg, placing it in the lowest toxicity category. The Cosmetic Ingredient Review (CIR) assessed acetyl hexapeptide-8 amide and found it safe as used in cosmetic formulations. Cytotoxicity studies show that significant cell damage requires concentrations 18- to 10,000-fold higher than those used in cosmetic products, depending on cell type.
Argireline is regulated as a cosmetic ingredient, not a drug. In the European Union, the maximum recommended use concentration in leave-on cosmetic products is 0.005% (50 ppm) of the active peptide. In the United States, the FDA does not regulate cosmetic ingredients with the same rigor as drugs. Argireline products can be sold without demonstrating clinical efficacy or safety through the FDA approval process, provided they do not make drug claims (such as claiming to treat a disease or alter the structure or function of the body).
This regulatory distinction matters for consumers. Claims about argireline's effects on wrinkles are cosmetic claims, not medical claims. They are not subject to the same evidentiary standards as pharmaceutical drug claims. The clinical trials described above are manufacturer-sponsored and do not meet the regulatory bar for drug approval.
Where the Evidence Stands
Argireline is a well-designed peptide with a clear molecular rationale, consistent in vitro activity, measurable clinical effects in controlled studies, and an excellent safety profile. The original Blanes-Mira et al. study demonstrated the concept: a peptide fragment of SNAP-25 can interfere with SNARE complex assembly and reduce neurotransmitter release.[1] The Wang et al. randomized trial confirmed that topical application produces statistically significant wrinkle reduction compared to placebo.[2]
The limitations are equally clear. Skin penetration is minimal (0.01% to the epidermis). No study has shown that topical argireline produces effects comparable to injectable botulinum toxin. The mechanism by which topical argireline reduces wrinkles may not be the SNARE complex inhibition demonstrated in cell culture. The clinical trials are small, manufacturer-sponsored, and use instrumental measurements rather than standardized clinical grading scales. Independent, large-scale trials with standardized endpoints are absent.
Argireline occupies a legitimate but narrow space in skincare: a peptide that produces modest, measurable improvement in skin surface texture with virtually no risk. The "Botox in a bottle" label overpromises. But dismissing argireline entirely would also be wrong. The clinical evidence, while limited, is consistent and positive. For consumers seeking a non-injectable option with realistic expectations, argireline remains the most studied neurotransmitter-inhibiting cosmetic peptide available.
The Bottom Line
Argireline (acetyl hexapeptide-8) is a synthetic 6-amino-acid peptide designed to mimic the N-terminus of SNAP-25 and destabilize the SNARE complex required for neurotransmitter release. In vitro, it reduces acetylcholine release with measurable potency. In clinical trials, topical application reduced wrinkle depth by 17-30% after 15-30 days in the original study, and showed 48.9% total anti-wrinkle efficacy versus 0% for placebo in a randomized controlled trial. The principal limitation is skin penetration: only 0.01% of applied argireline reaches the epidermis due to its 889 Da molecular weight and hydrophilic character. Effects are real but modest compared to injectable botulinum toxin. The safety profile is excellent, with LD50 over 2,000 mg/kg.
Sources & References
- 1RPEP-00716·Blanes-Mira, C et al. (2002). “Argireline: The Anti-Wrinkle Peptide Designed to Mimic Botox Without the Needles.” International journal of cosmetic science.Study breakdown →PubMed →↩
- 5RPEP-02768·Pickart, Loren et al. (2015). “GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration..” BioMed research international.Study breakdown →PubMed →↩
- 6RPEP-04623·Aguirre-Cruz, Gabriel et al. (2020). “Collagen Peptides for Skin: What Oral Supplements and Topical Products Actually Do.” Antioxidants (Basel.Study breakdown →PubMed →↩
- 7RPEP-05306·Chae, Minjung et al. (2021). “Fish Collagen Peptides Protect Skin Collagen From Stress Hormone Damage.” International journal of molecular sciences.Study breakdown →PubMed →↩