Amphibian Skin Peptides: A Frog's Pharmacy
Venom and Animal-Derived Peptides
3,000+ Peptides Discovered
More than 3,000 bioactive peptides have been isolated from amphibian skin secretions. These include antimicrobials, neuropeptides, insulin-releasing compounds, and opioid receptor ligands. One family, the magainins, reached Phase III clinical trials.
Zasloff, PNAS, 1987
Zasloff, PNAS, 1987
If you only read one thing
Frog skin is basically a living pharmacy. Scientists have found over 3,000 different germ-killing and brain-active peptides in the skin of frogs, toads, and salamanders. Some kill antibiotic-resistant bacteria. Others led to major discoveries in brain science. But after nearly 40 years of research, not a single one has been approved as a human drug — the closest attempt, a cream for infected foot ulcers, failed its final clinical trial twice.
In 1987, Michael Zasloff noticed something that surgical textbooks could not explain. African clawed frogs (Xenopus laevis) in his laboratory at the National Institutes of Health recovered from surgery without infection, despite being housed in non-sterile water teeming with bacteria. He investigated their skin and isolated two 23-amino-acid peptides with broad-spectrum antimicrobial activity against bacteria, fungi, and protozoa.[1] He named them magainins, from the Hebrew word for shield. That discovery launched a field. In the four decades since, researchers have identified over 3,000 bioactive peptides from the skin secretions of frogs, toads, salamanders, and caecilians. For the broader story of how animal venoms and secretions produce drug candidates, see Venom Peptides: How Deadly Toxins Become Life-Saving Drugs.
Key Takeaways
- Frog skin is basically a living pharmacy. Over 3,000 different bioactive peptides have been pulled out of the skin of frogs, toads, and salamanders.
- The discovery started with a mystery: a scientist noticed his lab frogs never got infected, even in dirty water. The answer was in their skin.
- These peptides kill bacteria by punching holes in their outer membrane — a completely different approach than standard antibiotics use.
- Because the attack hits the whole membrane at once, bacteria can't evolve resistance the way they do to conventional antibiotics. That's the hook.
- After nearly 40 years of research, zero frog-skin peptides have been approved as human drugs. The closest, a foot-ulcer cream, failed its final trial twice.
- Frog skin has also unlocked brain chemistry. Two mammalian neurotransmitters were discovered only after scientists first found them in frogs.
- Beyond infections, frog-skin peptides trigger insulin release, activate opioid receptors, and even heal wounds — a genuinely untapped drug pipeline.
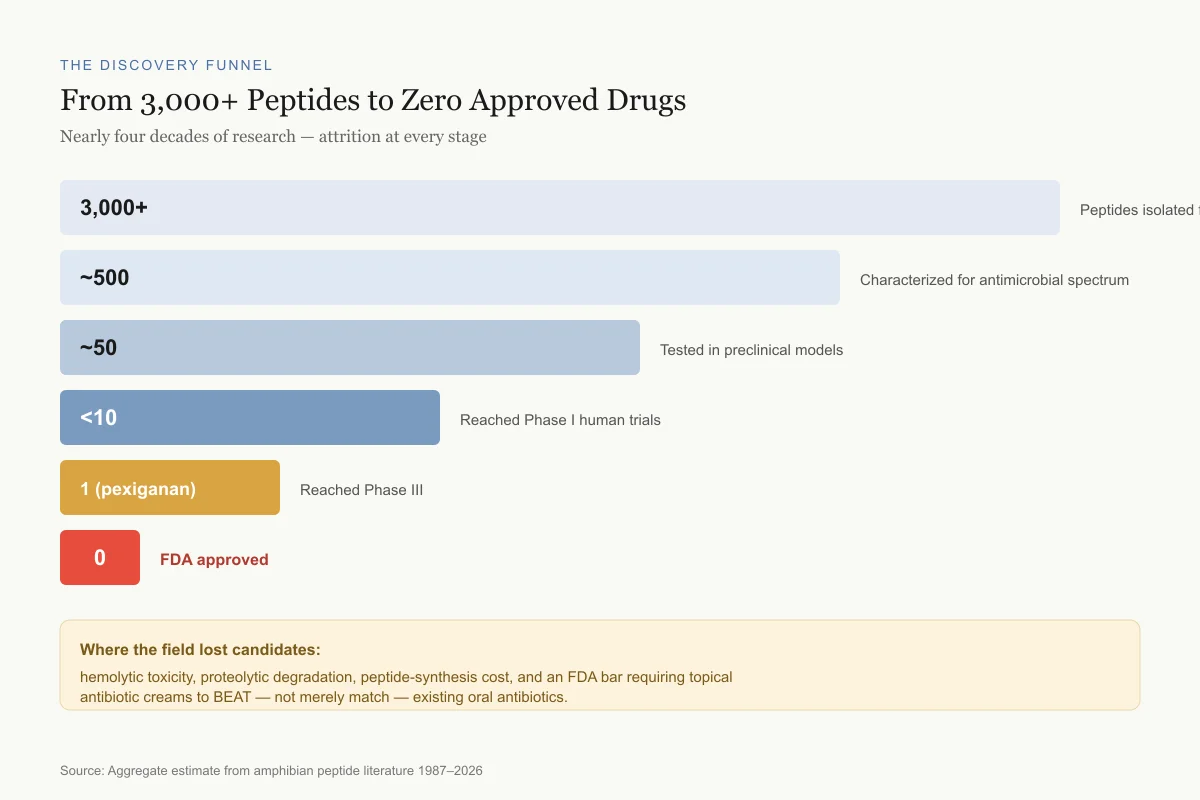
The Discovery Funnel
From 3,000+ Peptides to Zero Approved Drugs
Nearly four decades of research, attrition at every stage
3,000+
Peptides isolated from amphibian skin
~500
Characterized for antimicrobial spectrum
~50
Tested in preclinical models
<10
Reached Phase I human trials
1 (pexiganan)
Reached Phase III
0
FDA approved
Where the field lost candidates: hemolytic toxicity, proteolytic degradation, peptide-synthesis cost, and an FDA bar requiring topical antibiotic creams to beat, not merely match, existing oral antibiotics.
Source: Aggregate estimate from amphibian peptide literature 1987–2026
 View as image
View as imageWhy Frogs Make Antimicrobial Peptides
Amphibians lack the keratinized skin barrier that protects mammals from environmental pathogens. A frog's skin is thin, moist, and permeable, designed for gas exchange and water absorption. This permeability makes it vulnerable to infection by bacteria, fungi, and parasites present in aquatic and terrestrial habitats.
To compensate, amphibian skin contains specialized granular glands (also called poison glands or serous glands) that synthesize, store, and secrete a complex cocktail of bioactive peptides on demand. When stressed or injured, the frog releases these peptides onto its skin surface, creating a chemical barrier against microbial invasion. Rollins-Smith (2005) described this system as the amphibian equivalent of an innate immune defense, operating at the skin surface before pathogens can establish infection.[2]
The diversity of amphibian skin peptides is extraordinary. A single species may produce 10 to 30 distinct peptides, each with different antimicrobial spectra, mechanisms, and potencies. Different frog families produce different peptide families: Xenopus produces magainins and PGLa, Phyllomedusa tree frogs produce dermaseptins, European Rana frogs produce temporins and brevinins, and Bombina toads produce bombinins. This chemical diversity represents hundreds of millions of years of evolutionary optimization against microbial pathogens.
The peptides are synthesized as inactive prepropeptides and stored in granules within the gland cells. Upon adrenergic stimulation (triggered by stress, injury, or predator contact), the granules release their contents onto the skin surface, where enzymatic processing produces the active, mature peptides. This system allows rapid deployment: a frog can flood its skin surface with antimicrobial peptides within seconds of detecting a threat. The concentration of peptides achieved on the skin surface during secretion reaches levels far above the minimum inhibitory concentration (MIC) for most environmental pathogens.
The Major Peptide Families
Magainins
Magainins remain the best-studied amphibian antimicrobial peptides. Zasloff isolated magainin-1 and magainin-2 from Xenopus laevis skin in 1987.[1] Both are 23 amino acids long and differ by two substitutions. They adopt an alpha-helical conformation in the presence of bacterial membranes and kill through a mechanism now understood to involve toroidal pore formation.
A 2026 study by Zan et al. in PLoS Pathogens used molecular dynamics simulations to demonstrate that PGLa (peptide glycine-leucine-amide, another Xenopus peptide) and magainin-2 form transient hourglass-shaped toroidal pores in bacterial membranes.[3] These pores are short-lived but cause catastrophic membrane disruption. PGLa and magainin-2 also work synergistically: when combined, they kill bacteria at concentrations where neither is effective alone. This synergy is among the strongest observed for any antimicrobial peptide pair and has attracted interest for combination therapy strategies.
Saad et al. (2025) characterized the structure and dynamics of magainin-2 in biomimetic membranes, providing atomic-level detail of how the peptide transitions from a surface-bound state to a membrane-inserted state during pore formation.[4]
Dermaseptins
Dermaseptins were first isolated from the skin of South American tree frogs in the genus Phyllomedusa. They are 24 to 34 amino acids long, cationic, and adopt amphipathic alpha-helical structures similar to magainins. Their antimicrobial spectrum covers Gram-negative bacteria, Gram-positive bacteria, fungi, and viruses, including in vitro activity against HIV.
A 2026 study by Haddad et al. tested peptide derivatives of dermaseptins against multidrug-resistant bacteria, demonstrating activity against clinical isolates that resist conventional antibiotics.[5] The researchers modified the natural dermaseptin sequence to improve stability and reduce hemolytic toxicity, a common challenge with amphibian peptides. For more on how these principles apply to other venomous creatures, see Snake Venom Peptides in Medicine: From Bite to Breakthrough.
Temporins
Temporins are among the shortest amphibian antimicrobial peptides, typically 10 to 14 amino acids. First isolated from the European red frog Rana temporaria, they are the smallest alpha-helical antimicrobial peptides found in nature. Their small size makes them attractive for drug design because synthesis is cheaper and they are easier to modify.
Bellavita et al. (2021) designed the first cyclic temporin-L analogue, improving both antimicrobial activity and proteolytic stability compared to the linear parent peptide.[6] Jin et al. (2026) showed that temporin-derived peptides promote MRSA-infected wound healing in mice while protecting against secondary infection, demonstrating dual antimicrobial and wound-healing properties.[7]
Zannella et al. (2026) used structure-guided design of temporin derivatives to create peptides with potent dual-mechanism activity against both bacteria and biofilms.[8]
Other Families
The diversity extends beyond these three major groups. Brevinins and esculentins from Rana species show potent antimicrobial and anticancer activity. Bombinins from Bombina toads include both L-amino acid and D-amino acid forms, making them unusual among natural peptides. Phylloseptins from Phyllomedusa frogs show hepatic safety profiles that make them candidates for systemic use. Caerulein and xenopsin, originally identified as neuroactive peptides in frog skin, turned out to be analogs of mammalian neuropeptides, a discovery that reshaped neuroscience.
Bonilla-Jimenez et al. (2025) isolated and engineered a novel antimicrobial peptide from the skin of the Agua Rica leaf frog, demonstrating that new amphibian species continue to yield new peptide scaffolds for drug design.[9]
Beyond these major families, the field continues to expand. Pereira et al. (2025) published a proteomic and peptidomic characterization of the toad Rhaebo guttatus, identifying dozens of novel peptides with antimicrobial, anti-inflammatory, and antioxidant activities from a single species. The sheer number of unexplored amphibian species (over 8,000 named species, many never screened) suggests that the current catalogue of 3,000+ peptides represents a fraction of what exists in nature.
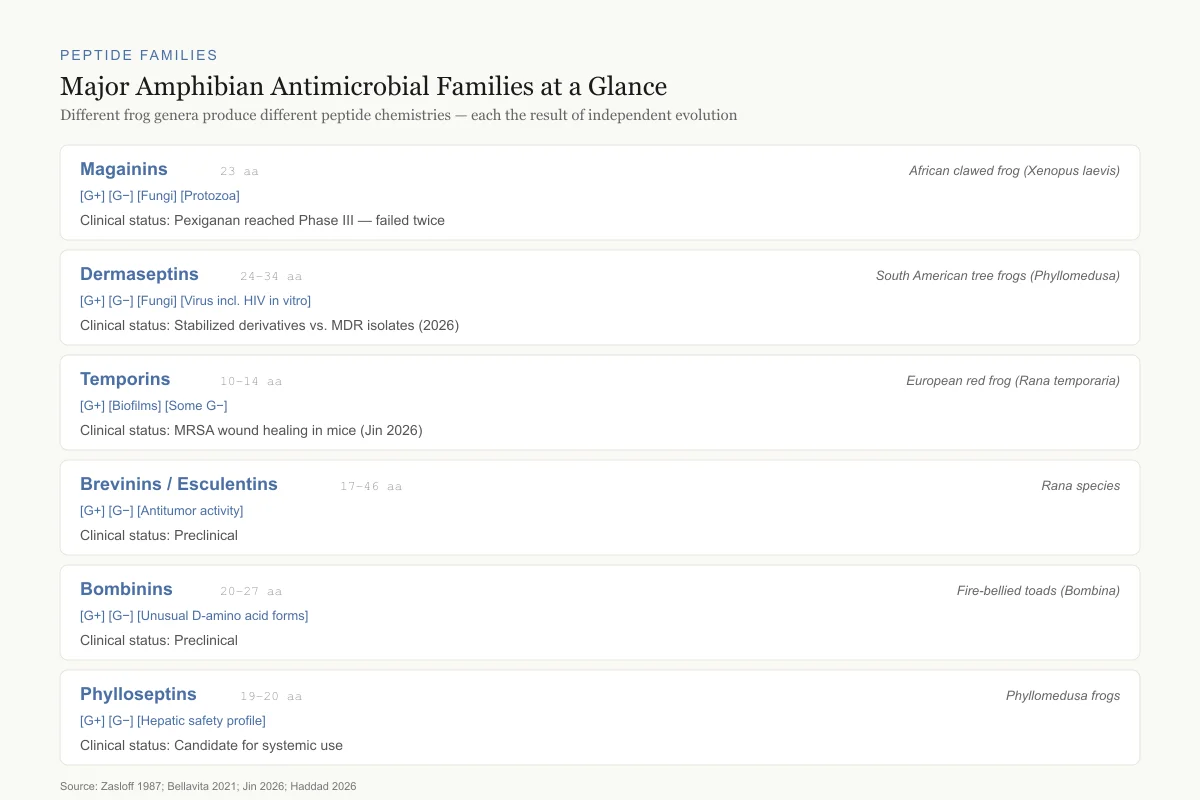
Peptide Families
The Major Amphibian Antimicrobial Families at a Glance
Different frog genera produce different peptide chemistries — hundreds of millions of years of independent evolution
Magainins
23 aa
African clawed frog (Xenopus laevis)
Clinical status: Pexiganan reached Phase III — failed twice
Dermaseptins
24–34 aa
South American tree frogs (Phyllomedusa)
Clinical status: Stabilized derivatives vs. MDR isolates (2026)
Temporins
10–14 aa
European red frog (Rana temporaria)
Clinical status: MRSA wound healing in mice (Jin 2026)
Brevinins / Esculentins
17–46 aa
Rana species
Clinical status: Preclinical
Bombinins
20–27 aa
Fire-bellied toads (Bombina)
Clinical status: Preclinical
Phylloseptins
19–20 aa
Phyllomedusa frogs
Clinical status: Candidate for systemic use
Source: Zasloff 1987; Bellavita 2021; Jin 2026; Haddad 2026
 View as image
View as imageHow Amphibian Peptides Kill
The killing mechanism shared by most amphibian antimicrobial peptides involves electrostatic attraction to anionic bacterial membranes, followed by alpha-helix formation and membrane disruption. The specifics vary by peptide family.
Yakobi et al. (2025) published a structural comparison of HNP-1 (a human alpha-defensin), magainin-2, and LL-37 (a cathelicidin), showing that while all three kill bacteria through membrane disruption, each uses a distinct mechanism: magainin-2 forms toroidal pores, HNP-1 uses a carpet model, and LL-37 creates toroidal pores of a different geometry.[10] This mechanistic diversity means amphibian peptides offer templates for drug design that are distinct from defensin-based or cathelicidin-based approaches.
Roy et al. (2019) compared the antiviral activity of temporin-A and other frog-skin peptides against herpes simplex virus, influenza A, and hepatitis C, finding broad-spectrum antiviral activity through viral envelope disruption.[11]
A key advantage of membrane-targeting antimicrobial peptides is the difficulty bacteria face in developing resistance. Bacteria would need to fundamentally alter their membrane composition to evade peptide binding, an adaptation that carries severe fitness costs. Some bacteria have evolved partial resistance mechanisms, including modification of lipopolysaccharide (LPS) charge to reduce peptide binding and production of outer membrane proteases that degrade antimicrobial peptides before they reach the cytoplasmic membrane. But these adaptations are metabolically expensive and typically do not confer complete resistance. This property makes amphibian peptides of particular interest in the context of rising antibiotic resistance, where the WHO has identified antimicrobial resistance as one of the top global health threats.
The synergy between different amphibian peptides adds another dimension. In nature, frogs secrete multiple peptide families simultaneously. Magainin-2 and PGLa from Xenopus work synergistically, as do different members of the temporin family. Combination approaches inspired by this natural strategy may overcome the limitations of single-peptide therapies.
Beyond Antimicrobials: The Neuropeptide Connection
Amphibian skin peptides are not limited to antimicrobial defense. Several of the most important discoveries in neuropeptide biology originated from frog skin.
Caerulein, isolated from the skin of the Australian frog Litoria caerulea, turned out to be a structural analog of cholecystokinin (CCK), a mammalian gut hormone that regulates digestion and appetite. This cross-species discovery in the 1960s helped researchers identify the CCK receptor system in mammals.
Bombesin, isolated from the skin of the European fire-bellied toad Bombina bombina, led to the discovery of its mammalian counterpart, gastrin-releasing peptide (GRP). GRP receptors are overexpressed in several human cancers, and bombesin-based peptides are now used as targeting vectors for cancer imaging and drug delivery. Jozi et al. (2026) developed novel gallium-68-labeled bombesin derivatives for PET imaging of GRP receptor-positive tumors.[12] This cancer imaging application represents one of the most direct clinical translations of an amphibian skin peptide to date.
Dermorphin and deltorphin, isolated from Phyllomedusa frogs, are potent opioid receptor agonists that bind mu and delta opioid receptors, respectively. Dermorphin is 30 to 40 times more potent than morphine on a molar basis. These peptides contain D-amino acids (specifically D-alanine), which is extremely rare in vertebrate biology and initially caused skepticism about their authenticity. The D-amino acid confers resistance to enzymatic degradation and is essential for opioid receptor binding. Dermorphin was later found to be used as an illegal performance-enhancing drug in horse racing, where its potent analgesic effects masked pain in injured racehorses, leading to its prohibition by racing authorities.
Insulin-releasing peptides from Xenopus species stimulate insulin secretion from pancreatic beta cells through pathways distinct from existing incretin drugs. Caerulein-related peptides and xenopsin-related peptides from Xenopus borealis and Xenopus amieti demonstrate significant insulin-releasing activity at nanomolar concentrations. Yuan et al. (2023) developed a potent Xenopus GLP-1-based triple agonist targeting GLP-1, glucagon, and Y2 receptors, demonstrating that frog-derived peptide scaffolds can be engineered into metabolic drug candidates.[13] Liu et al. (2025) further developed a Xenopus GLP-1-derived GLP-1/GIP/Y2 receptor tri-agonist with extended duration of action, building on the frog peptide scaffold to create a candidate for obesity and type 2 diabetes treatment.[15]
Bradykinin-related peptides represent another category found in amphibian skin. These peptides affect blood pressure, smooth muscle contraction, and pain signaling. Their discovery in frog skin preceded and facilitated the identification of mammalian bradykinin pathways, following the same pattern as caerulein and bombesin: frog skin peptide discovered first, mammalian homolog identified after.
Frog → Human Biology
Frog Skin Peptides That Reshaped Human Medicine
Each of these amphibian discoveries led to identification of a mammalian counterpart with major clinical impact
Amphibian peptide
Caerulein
Australian tree frog (Litoria caerulea)
Mammalian homolog
Cholecystokinin (CCK)
Led to identification of the CCK receptor system — regulates digestion and appetite
Amphibian peptide
Bombesin
European fire-bellied toad (Bombina bombina)
Mammalian homolog
Gastrin-Releasing Peptide (GRP)
GRP receptors overexpressed in cancers; bombesin tracers used in PET imaging today
Amphibian peptide
Dermorphin / Deltorphin
Phyllomedusa tree frogs
Mammalian homolog
Opioid receptor ligands (μ and δ)
30–40× more potent than morphine; contains rare natural D-amino acids
Amphibian peptide
Bradykinin-related peptides
Multiple frog species
Mammalian homolog
Bradykinin pathway
Frog-skin discovery preceded and enabled identification of mammalian bradykinin biology
Amphibian peptide
Xenopus GLP-1 analogs
African clawed frogs (Xenopus)
Mammalian homolog
GLP-1 / GIP / Y2 multi-agonist candidates
Engineered into tri-agonist candidates for obesity and type 2 diabetes (Yuan 2023; Liu 2025)
Source: Historical amphibian peptide discoveries; Yuan 2023; Liu 2025
 View as image
View as imageThe Drug Development Gap
Despite four decades of research and thousands of characterized peptides, no amphibian skin peptide has been approved as a human drug. The closest attempt was pexiganan (MSI-78), a synthetic analog of magainin-2.
Pexiganan was developed as a topical cream for infected diabetic foot ulcers by Genaera Corporation (formerly Magainin Pharmaceuticals, the company Zasloff founded). It progressed through Phase I and Phase II trials showing antimicrobial activity and wound healing benefits. In Phase III trials conducted in the late 1990s, pexiganan was as effective as oral ofloxacin (a fluoroquinolone antibiotic) in resolving diabetic foot infections, but it failed to demonstrate statistical superiority. The FDA declined approval on the basis that a topical peptide cream should outperform, not merely match, an established oral antibiotic.
Dipexium Pharmaceuticals later acquired the rights and initiated a second Phase III trial program in the 2010s. These trials also failed to demonstrate superiority over standard care. The pexiganan program represents over $100 million in development costs across two decades without reaching market.
The failure was not due to lack of antimicrobial activity. Pexiganan killed bacteria effectively in vitro and in wound models. The problem was clinical trial design and the high bar for demonstrating benefit in a complex wound healing population where infection is one factor among many influencing outcomes. Whether a different trial design, indication, or delivery method could have succeeded remains debated.
The pexiganan story illustrates the core challenge for amphibian peptide drug development. In vitro activity does not automatically translate to clinical superiority. Peptide stability, formulation, delivery, cost of manufacturing, and regulatory bar all create attrition. Several limitations recur across the field:
Hemolytic toxicity. Many amphibian antimicrobial peptides damage mammalian red blood cells at concentrations close to their antimicrobial effective dose. The therapeutic window between killing bacteria and killing human cells is often narrow.
Proteolytic degradation. Natural peptides are rapidly degraded by proteases in human blood and tissue. Cyclic analogs and D-amino acid substitutions can improve stability, as demonstrated by Bellavita et al. (2021) with cyclic temporin-L.[6]
Cost of synthesis. Solid-phase peptide synthesis for 20+ amino acid peptides remains expensive compared to small-molecule antibiotics. Shorter peptides like temporins (10-14 amino acids) have an economic advantage.
Regulatory path. Antimicrobial peptides occupy an uncertain regulatory category. They are not traditional small-molecule antibiotics, not biologics in the conventional sense, and not easily classified as medical devices when used topically.
Selectivity. The therapeutic index, the ratio between the concentration that kills bacteria and the concentration that damages host cells, varies widely among amphibian peptides. Magainin-2 has a relatively favorable therapeutic index, which is why it progressed to clinical trials. Many other amphibian peptides have indices too narrow for systemic use, restricting them to topical applications at best. Engineering efforts focus on improving this ratio through sequence modifications, cyclization, and non-natural amino acid incorporation.
Amphibian Decline and Bioprospecting
There is an ironic tension in amphibian peptide research. Frogs are simultaneously one of the richest sources of antimicrobial drug candidates and one of the most threatened vertebrate groups on the planet. Over 40% of amphibian species are declining, driven by habitat loss, climate change, and the chytrid fungus Batrachochytrium dendrobatidis (Bd). Bd attacks the skin, the very organ that produces the defensive peptides. Species with weaker antimicrobial peptide defenses are more susceptible to chytrid-driven extinction.
Fan et al. (2025) demonstrated that a frog skin-derived antimicrobial peptide suppresses atherosclerosis by modulating macrophage polarization, shifting macrophages from a pro-inflammatory M1 phenotype to an anti-inflammatory M2 phenotype.[14] This cardiovascular application demonstrates that frog skin peptides have therapeutic potential beyond antimicrobial use, paralleling findings in other peptide fields where antimicrobial peptides show anti-inflammatory, immunomodulatory, and tissue-repair properties.
Each species lost represents a unique peptide library that cannot be recovered. The chytrid fungus has already driven multiple frog species to extinction, and the peptide profiles of these species were never characterized. Conservation of amphibian biodiversity is inseparable from the future of amphibian-derived drug discovery. Organizations like the Amphibian Survival Alliance have begun cataloging skin secretion profiles from at-risk species specifically to preserve pharmaceutical potential.
Where the Field Stands
Amphibian skin peptides opened the modern era of antimicrobial peptide research. Zasloff's 1987 magainin paper has been cited over 4,000 times. The peptide families discovered since then, dermaseptins, temporins, brevinins, bombinins, and dozens of others, have provided structural templates, mechanistic insights, and drug design principles that extend beyond amphibians into the broader antimicrobial peptide field.
The approved drug count remains at zero. But the field has shifted from discovery (finding new peptides) toward engineering (modifying known peptides for clinical use). Cyclic temporins, stabilized dermaseptin derivatives, and bombesin-targeted imaging agents represent a maturation from natural product cataloging to rational drug design. For related approaches from other animal sources, see Bee Venom Peptides: Melittin and Apamin in Research, Cone Snail Peptides (Conotoxins), and Scorpion Venom Peptides: Chlorotoxin and Cancer Imaging.
The Bottom Line
Amphibian skin secretions have yielded over 3,000 bioactive peptides across antimicrobial, neuropeptide, and metabolic categories. Magainins, dermaseptins, and temporins kill bacteria through membrane disruption, a mechanism that resists conventional resistance development. No amphibian peptide has been approved as a drug; pexiganan (a magainin analog) failed Phase III for diabetic foot ulcers. Current research focuses on engineering stabilized analogs, with 2026 studies showing temporin derivatives healing MRSA-infected wounds and dermaseptin derivatives active against multidrug-resistant bacteria.
Sources & References
- 1RPEP-00063·Zasloff, M (1987). “Magainins: The Frog Skin Peptides That Kill Bacteria, Fungi, and Parasites.” Proceedings of the National Academy of Sciences of the United States of America.Study breakdown →PubMed →↩
- 2RPEP-01080·Rollins-Smith, Louise A et al. (2005). “Antimicrobial Peptide defenses in amphibian skin..” Integrative and comparative biology.Study breakdown →PubMed →↩
- 3RPEP-16528·Zan, Bing et al. (2026). “PGLa and Magainin 2 Can Porate Membranes via Transient Hourglass-Shaped Toroidal Pores..” Journal of the American Chemical Society.Study breakdown →PubMed →↩
- 4RPEP-13346·Saad, Ahmad et al. (2025). “Structure and Dynamics of the Magainin 2 Antimicrobial Peptide in Biomimetic Lipid Bilayers by Solid-State NMR..” Biochemistry.Study breakdown →PubMed →↩
- 5RPEP-15248·Haddad, Houda et al. (2026). “Dermaseptin Peptide Derivatives Kill MDR Klebsiella and Staph at Low Concentrations.” Biochemistry and biophysics reports.Study breakdown →PubMed →↩
- 6RPEP-05276·Bellavita, Rosa et al. (2021). “First Cyclic Version of Temporin L Antimicrobial Peptide Designed With Enhanced Helical Structure.” Journal of medicinal chemistry.Study breakdown →PubMed →↩
- 7RPEP-15387·Jin, Xiao et al. (2026). “Frog-Derived Temporin Peptides Heal MRSA Wounds and Protect Against Lethal Sepsis in Mice.” Bioorganic chemistry.Study breakdown →PubMed →↩
- 8RPEP-16530·Zannella, Carla et al. (2026). “Structure-Guided Design of Temporin-Derived Peptides Reveals Potent Dual-Mechanism Inhibitors of SARS-CoV-2..” Journal of medicinal chemistry.Study breakdown →PubMed →↩
- 9RPEP-10197·Bonilla-Jiménez, Stefanny et al. (2025). “Ecuadorian Frog Skin Yields a Peptide That Engineers Into a Potent Antibiotic.” Antibiotics (Basel.Study breakdown →PubMed →↩
- 10RPEP-14295·Yakobi, Sinethemba H et al. (2025). “Structural and mechanistic divergence in LL-37, HNP-1, and Magainin-2: An integrated computational and biophysical analysis..” Current research in structural biology.Study breakdown →PubMed →↩
- 11RPEP-04453·Roy, Maëva et al. (2019). “Comparison of Anti-Viral Activity of Frog Skin Anti-Microbial Peptides Temporin-Sha and [K³]SHa to LL-37 and Temporin-Tb against Herpes Simplex Virus Type 1..” Viruses.Study breakdown →PubMed →↩
- 12RPEP-15394·Jozi, Shireen et al. (2026). “Novel 68Ga-Labeled Peptide PET Tracers Target Gastrin-Releasing Peptide Receptor for Cancer Imaging.” Molecular pharmaceutics.Study breakdown →PubMed →↩
- 13RPEP-07604·Yuan, Yongliang et al. (2023). “Discovery of a potent and long-acting Xenopus GLP-1-based GLP-1/glucagon/Y2 receptor triple agonist..” European journal of medicinal chemistry.Study breakdown →PubMed →↩
- 14RPEP-10888·Fan, Fan et al. (2025). “A Frog Skin Antimicrobial Peptide Reduces Atherosclerotic Plaque Formation by Suppressing Inflammation Through a Novel Molecular Pathway.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 15RPEP-12251·Liu, Xiyu et al. (2025). “Development of a potent Xenopus GLP-1-derived GLP-1/GIP/Y2 receptor tri-agonist for obesity and type 2 diabetes..” International journal of biological macromolecules.Study breakdown →PubMed →↩