AMH: The Fertility Biomarker Peptide Explained
Reproductive Peptides
Best ovarian reserve marker
AMH outperforms FSH, estradiol, and inhibin B for predicting ovarian response to IVF stimulation, with the advantage of minimal cycle-to-cycle variation.
Broer et al., Hum Reprod Update, 2014
Broer et al., Hum Reprod Update, 2014
If you only read one thing
AMH is a blood test that tells you roughly how many eggs you have left. It's the best test we have for that purpose — better than FSH, and you can take it on any day of your cycle. But here's the thing most people get wrong: AMH tells you about egg quantity, not egg quality. A low AMH doesn't mean you can't get pregnant. It means you'd likely produce fewer eggs in an IVF cycle. Age, not AMH, is the biggest factor in whether those eggs lead to a baby.
Key Takeaways
- AMH is a blood test that roughly estimates how many eggs you have left — think of it as a balance check on your ovarian savings account.
- Unlike most fertility tests, you can take it on any day of your cycle. AMH levels barely move month to month, which is part of why it's the best reserve marker we have.
- The biggest misconception: low AMH doesn't mean you can't get pregnant. It means fewer eggs would likely be retrieved in an IVF cycle — not that each egg is worse.
- Egg quality is age-dependent, not AMH-dependent. A 32-year-old with low AMH has the same-quality eggs as any other 32-year-old.
- AMH is also a PCOS marker. Women with PCOS run 2 to 3 times higher than average because they have an excess of small follicles.
- It can roughly predict when menopause will arrive, but not by much — adding AMH to age only improves the estimate by a couple of percentage points.
- Doctors warn against using AMH as a general fertility barometer for women not actively trying to conceive. It's a guide for treatment, not a verdict.
What Is Anti-Mullerian Hormone?
AMH is a 140-kilodalton glycoprotein belonging to the transforming growth factor-beta (TGF-beta) superfamily. It was first identified for its role in male fetal development: AMH produced by the fetal testes causes the Mullerian ducts (which would otherwise develop into the uterus, fallopian tubes, and upper vagina) to regress. This is where the name comes from: anti-Mullerian, meaning it opposes the development of Mullerian structures.[2]
In women, AMH plays a completely different role. It is produced by granulosa cells of small growing follicles in the ovary, specifically pre-antral and small antral follicles. AMH acts as a brake on follicle recruitment. It inhibits the initial recruitment of primordial follicles into the growing pool and reduces the sensitivity of growing follicles to FSH.[1] Without AMH, too many primordial follicles would activate at once, depleting the reserve prematurely.
Dewailly et al. (2014) published a comprehensive review of AMH physiology in Human Reproduction Update. They documented that AMH is produced exclusively by granulosa cells and is independent of gonadotropin stimulation. This gonadotropin independence is the key property that makes AMH a superior biomarker: its levels do not fluctuate with the menstrual cycle the way FSH and estradiol do.[2]
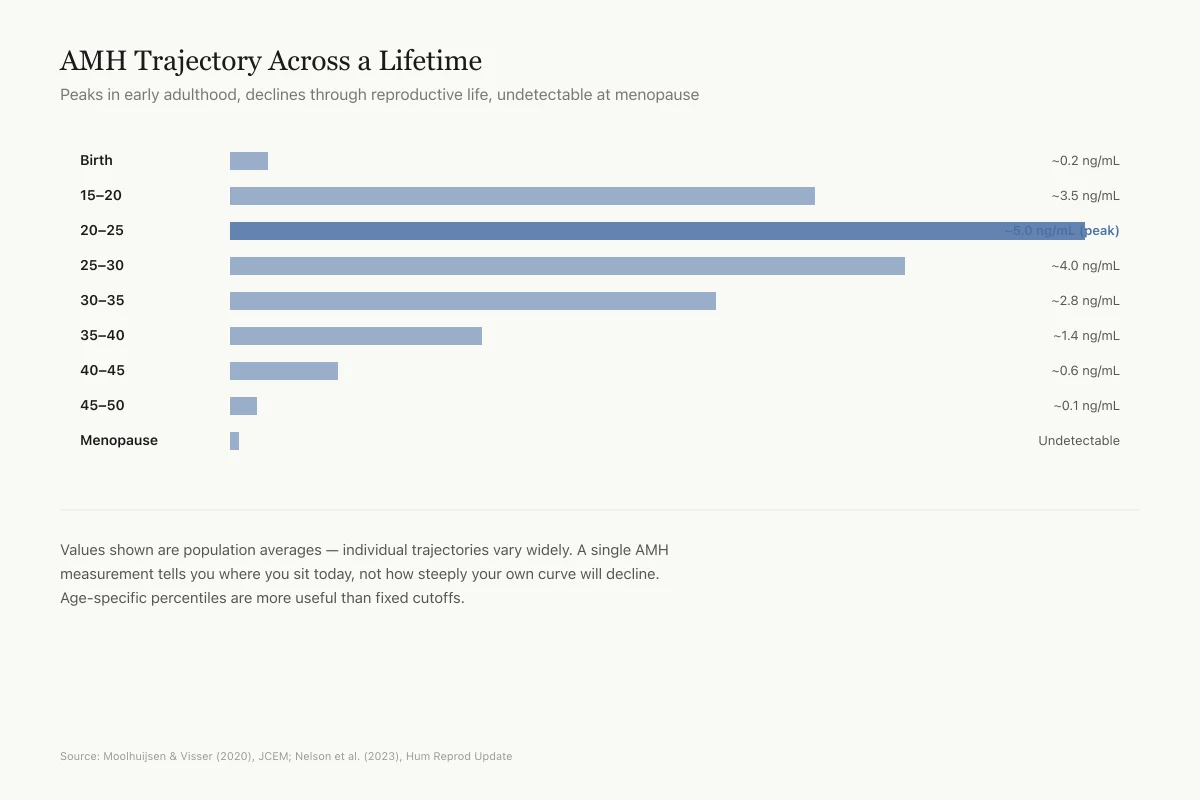
AMH levels are highest in early adulthood (around ages 20-25), decline gradually through the 30s, drop more steeply after 35, and become undetectable around menopause. This trajectory mirrors the decline in the number of remaining follicles, making AMH a real-time readout of the ovarian reserve.
AMH Across a Lifetime
Approximate Trajectory of AMH Levels
Peaks in early adulthood, declines throughout reproductive life, undetectable at menopause
Birth
~0.2 ng/mL
15–20
~3.5 ng/mL
20–25
~5 ng/mL
25–30
~4 ng/mL
30–35
~2.8 ng/mL
35–40
~1.4 ng/mL
40–45
~0.6 ng/mL
45–50
~0.1 ng/mL
Menopause
~0 ng/mL
Values shown are population averages — individual trajectories vary widely. A single AMH measurement tells you where you sit today, not how steeply your own curve will decline. Age-specific percentiles are more useful than fixed cutoffs.
Source: Moolhuijsen & Visser (2020), JCEM; Nelson et al. (2023), Hum Reprod Update — population means
 View as image
View as imageAMH as an Ovarian Reserve Marker
Why AMH replaced FSH
For decades, day-3 FSH was the standard test for ovarian reserve. But FSH has fundamental problems as a biomarker. It only rises when the ovarian reserve is already very low, making it a late indicator. It varies throughout the menstrual cycle, requiring timed blood draws. And it is influenced by estrogen, body weight, and medications.[1]
Broer et al. (2014) published a landmark review in Human Reproduction Update concluding that AMH is the best currently available measure of ovarian reserve across multiple clinical contexts. They found that AMH outperformed FSH, estradiol, inhibin B, and antral follicle count (AFC) for predicting ovarian response to IVF stimulation, forecasting reproductive lifespan, and identifying ovarian dysfunction in PCOS.[1]
The advantages of AMH over FSH are straightforward:
- AMH can be measured on any day of the menstrual cycle
- AMH declines years before FSH rises, detecting diminished reserve earlier
- AMH has less cycle-to-cycle variability (though it is not perfectly constant)
- AMH correlates directly with the number of growing follicles
What AMH levels mean
Moolhuijsen and Visser (2020) published an updated review on AMH and ovarian function assessment. They noted that while specific cutoff values vary by assay and laboratory, general interpretive ranges are:[4]
- Below 1.0 ng/mL: Diminished ovarian reserve. Fewer eggs remain. Lower expected response to IVF stimulation.
- 1.0-3.5 ng/mL: Normal range for most reproductive-age women. Expected adequate IVF response.
- Above 3.5-5.0 ng/mL: High levels. Possible PCOS. Higher risk of ovarian hyperstimulation syndrome during IVF.
These ranges must be interpreted in the context of age. An AMH of 1.5 ng/mL in a 25-year-old is concerning; the same value in a 40-year-old is expected. Age-specific percentile charts have been developed to account for this.
A critical limitation: AMH measures the quantity of remaining eggs, not their quality. Egg quality is primarily determined by age. A 42-year-old with a normal AMH still has lower pregnancy rates than a 30-year-old with the same AMH, because her eggs are more likely to have chromosomal abnormalities.
AMH and IVF
Predicting oocyte yield
The single strongest clinical application of AMH is predicting how many eggs a woman will produce during IVF stimulation. La Marca et al. (2010) established AMH as a predictive marker in assisted reproductive technology, demonstrating a strong positive correlation between pre-treatment AMH levels and the number of oocytes retrieved.[5]
This allows clinicians to individualize FSH dosing. Women with low AMH receive higher doses to maximize egg retrieval. Women with high AMH receive lower doses to reduce the risk of ovarian hyperstimulation syndrome (OHSS), a potentially dangerous condition where the ovaries over-respond and cause fluid shifts, pain, and in rare cases blood clots.
Predicting live birth
The relationship between AMH and IVF pregnancy is weaker than the relationship between AMH and egg count. La Marca et al. (2011) developed an AMH-based prediction model for live birth after assisted reproduction. They found that AMH contributed to the prediction but that age remained the dominant factor.[6]
Granger and Tal (2019) reviewed the predictive utility of AMH in ART outcomes. Their conclusion was clear: AMH is an excellent predictor of ovarian response (how many eggs) but only a weak predictor of clinical pregnancy and live birth. The reason is that AMH measures follicle quantity, while pregnancy depends on egg quality, embryo development, uterine receptivity, and many other factors that AMH does not capture.[7]
This distinction matters because patients and sometimes clinicians conflate "low AMH" with "cannot get pregnant." Low AMH means fewer eggs will be retrieved in an IVF cycle, which does reduce the statistical probability of success per cycle. But individual eggs from women with low AMH are not inherently less capable of producing a pregnancy than eggs from women with normal AMH of the same age.
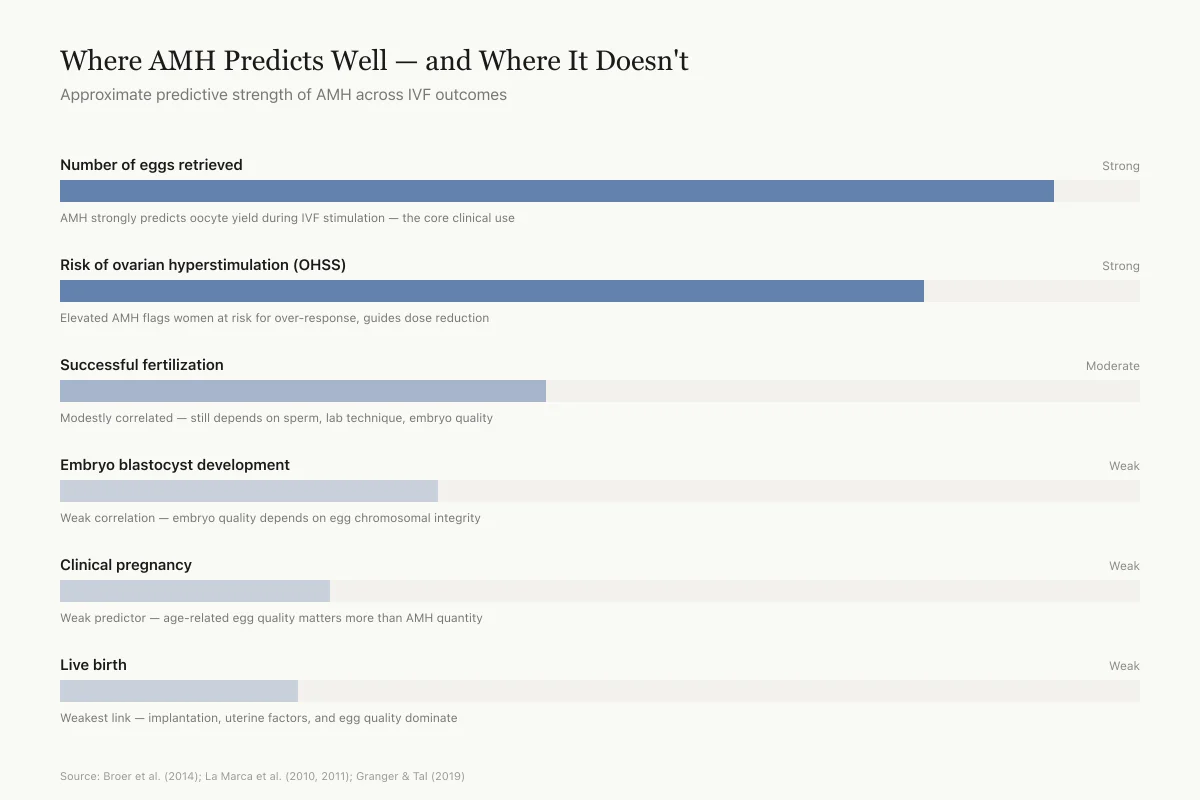
Quantity vs Quality
Where AMH Predicts Well — and Where It Doesn't
Approximate predictive strength of AMH across IVF outcomes
AMH strongly predicts oocyte yield during IVF stimulation — this is the core clinical use
Elevated AMH flags women at risk for over-response, guides dose reduction
Modestly correlated — still depends heavily on sperm, lab technique, embryo quality
Weak correlation — embryo quality depends on egg chromosomal integrity
Weak predictor — age-related egg quality matters more than AMH quantity
Weakest link — implantation, uterine factors, and egg quality dominate
AMH predicts how many eggs your ovaries will produce very well. It doesn't predict whether those eggs will turn into a baby — age does. Low AMH is a reason to plan, not a reason to panic.
Source: Broer et al. (2014); La Marca et al. (2010, 2011); Granger & Tal (2019) — synthesis of predictive value
 View as image
View as imageDosing individualization
One of the most practical applications of AMH in IVF is tailoring the dose of gonadotropin (FSH) medication used to stimulate the ovaries. The goal of IVF stimulation is to retrieve enough eggs for embryo selection without pushing the ovaries into dangerous hyperstimulation. AMH provides a pre-treatment estimate of how the ovaries will respond.
Women with low AMH (below 1.0 ng/mL) typically receive higher FSH doses because their ovaries respond weakly. Women with high AMH (above 3.5 ng/mL) receive lower doses because they are prone to excessive response. This AMH-guided dosing approach has been adopted by most IVF clinics worldwide and has reduced rates of both cancelled cycles (too few eggs) and OHSS (too many). La Marca et al. (2010) were among the first to formally establish the predictive framework for this dosing strategy.[5]
Egg freezing decisions
AMH testing has also influenced the growing field of elective egg freezing (oocyte cryopreservation). Women considering egg freezing for non-medical reasons often request AMH testing to understand whether their ovarian reserve is declining. A lower-than-expected AMH for age may prompt earlier action, while a reassuringly normal AMH may allow more time for decision-making.
The limitations apply here too: AMH tells women about egg quantity, not quality. A 38-year-old with excellent AMH still faces age-related egg quality challenges that AMH cannot measure. Egg freezing outcomes depend on both the number of eggs retrieved and the age at which they were frozen.
AMH and PCOS
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in reproductive-age women, affecting 6-12% of the population. A defining feature of PCOS is excess small antral follicles in the ovaries. Since these follicles produce AMH, women with PCOS have AMH levels 2-3 times higher than age-matched controls.[8]
Bhide and Homburg (2016) reviewed the relationship between AMH and PCOS in Human Reproduction Update. They found that elevated AMH reflects both the increased number of small follicles and the fact that each follicle in PCOS produces more AMH per cell than normal follicles. This dual mechanism explains why AMH elevations in PCOS can be dramatic.[8]
Multiple studies have proposed using AMH as a diagnostic criterion for PCOS, with cutoffs ranging from 3.8 to 5.0 ng/mL. The potential advantage is that a blood test is more objective and reproducible than ultrasound-based antral follicle counting, which depends on operator skill and equipment quality.
However, AMH has not been formally incorporated into the Rotterdam diagnostic criteria for PCOS. The international PCOS guideline (2023) acknowledged AMH's potential but stopped short of recommending it as a primary diagnostic tool, citing assay standardization issues and the need for more population-specific cutoff validation.
AMH and Menopause Prediction
If AMH tracks the decline of the ovarian reserve, can it predict when menopause will occur? This question has driven considerable research.
Nelson et al. (2023) published a systematic review in Human Reproduction Update evaluating AMH for menopause diagnosis and prediction. They found that lower age-specific AMH concentrations do predict a younger age at menopause. However, the practical utility is limited: adding AMH to age-based predictions improved the C-statistic from 84% to only 86%.[9]
The individual-level predictions were particularly imprecise. A woman with a low AMH at age 35 might reach menopause anywhere from age 42 to 52, a range so wide that it offers little actionable guidance. AMH is better at identifying women at the extremes (very early or very late menopause) than at predicting menopause timing for the average woman.
Women with PCOS represent a special case. Their elevated AMH levels correspond to a delayed menopause by approximately 4 years compared to regularly ovulating women. This aligns with the biology: more follicles take longer to deplete.
The American College of Obstetricians and Gynecologists has recommended against ordering AMH tests for women who are not seeking fertility treatment. The concern is that a "low" AMH result may cause unnecessary anxiety in women who are not trying to conceive, while a "normal" AMH may create false reassurance about fertility when age-related egg quality decline is the more important factor.
AMH and Cancer Treatment
AMH has found a clinical role in oncofertility, the field of preserving reproductive options for cancer patients. Chemotherapy and radiation can damage or destroy ovarian follicles. Because AMH reflects the number of remaining growing follicles, it drops rapidly during gonadotoxic treatment and can be used to track ovarian damage in real time.[10]
Pre-treatment AMH levels help predict the likelihood of post-chemotherapy ovarian recovery. Women with higher pre-treatment AMH are more likely to resume menstruation and retain fertility after treatment. Women with already low AMH before chemotherapy face a higher risk of permanent ovarian failure.
Post-treatment AMH monitoring can identify whether follicle growth has resumed. After alkylating agents (the most gonadotoxic chemotherapy class), AMH levels often drop to undetectable within weeks. If they recover over months or years, it suggests surviving follicles have re-entered the growth pool. If they remain undetectable, the follicle reserve has likely been destroyed.
This application extends beyond cancer. Any medical treatment or surgery that affects the ovaries (including endometriosis surgery, ovarian cyst removal, or ovarian drilling for PCOS) can reduce AMH levels. Pre- and post-procedure AMH testing helps quantify the impact on ovarian reserve.
At-Home AMH Testing
The emergence of direct-to-consumer AMH testing has expanded access but also created new clinical concerns. Companies now offer finger-prick AMH kits that patients can complete at home and mail to a laboratory. These tests use the same immunoassay technology as clinical-grade tests, and their accuracy for measuring AMH levels is generally comparable.[4]
Safety
ModerateAt-home AMH tests can mislead without clinical context
Concern
Direct-to-consumer AMH kits accurately measure the hormone level, but results without a doctor's interpretation can cause unnecessary panic or false confidence. A 'low' result in a 38-year-old may be completely normal for her age. A 'normal' result does not guarantee fertility because AMH says nothing about egg quality.
What the research says
Professional societies including ACOG recommend against using AMH to counsel women not actively seeking fertility treatment. If you take an at-home test, discuss the result with a reproductive endocrinologist who can interpret it in context of your age, goals, and health history.
Particularly relevant for: Women using direct-to-consumer fertility testing without clinical guidance
What to do
Never make major life decisions based on a single AMH number without professional interpretation. Context — especially your age — changes what the number means.
ACOG guidance; Moolhuijsen and Visser (2020), J Clin Endocrinol Metab
The concern is not test accuracy but test interpretation. Without clinical context, women may interpret results in misleading ways. A "low" AMH in a 38-year-old may be entirely normal for her age. A "normal" AMH does not guarantee fertility, because egg quality (which AMH does not measure) may already be declining. Professional organizations have expressed concern that at-home AMH testing outside the clinical setting may lead to unnecessary anxiety, false reassurance, or delayed appropriate fertility evaluation.
AMH in Men
AMH is not exclusively a female biomarker. In males, Sertoli cells in the testes produce AMH. Levels are high in boys and decline after puberty when testosterone suppresses AMH production. In men, AMH has been studied as a marker of Sertoli cell function and testicular development.[2]
Clinical applications of AMH in males include evaluating cryptorchidism (undescended testes) in boys, where detectable AMH indicates functioning testicular tissue, and assessing gonadal function in disorders of sex development. Russell et al. (2022) reviewed the clinical utilities of AMH, noting that male applications remain less developed than female ones but that AMH measurement can avoid more invasive diagnostic procedures in pediatric urology.[10]
Assay Standardization: An Ongoing Problem
One of the biggest obstacles to using AMH clinically is that different assays give different results for the same blood sample. The two most widely used AMH assays (Beckman Coulter Gen II and Roche Elecsys) produce values that are correlated but not interchangeable. A value of 2.0 ng/mL on one platform may not mean the same thing as 2.0 ng/mL on another.[4]
This matters because published cutoff values (for diminished reserve, PCOS diagnosis, or IVF dosing protocols) are assay-specific. Applying a cutoff derived from one assay to results from another can lead to misclassification. Efforts to establish an international standard reference preparation are underway but not yet complete.
Complement-based AMH (picoAMH) and automated platforms have improved reproducibility, but the lack of a universal standard remains a real limitation of AMH testing in clinical practice. Until a reference standard is adopted, clinicians and patients should always note which assay was used when interpreting results or comparing values over time.
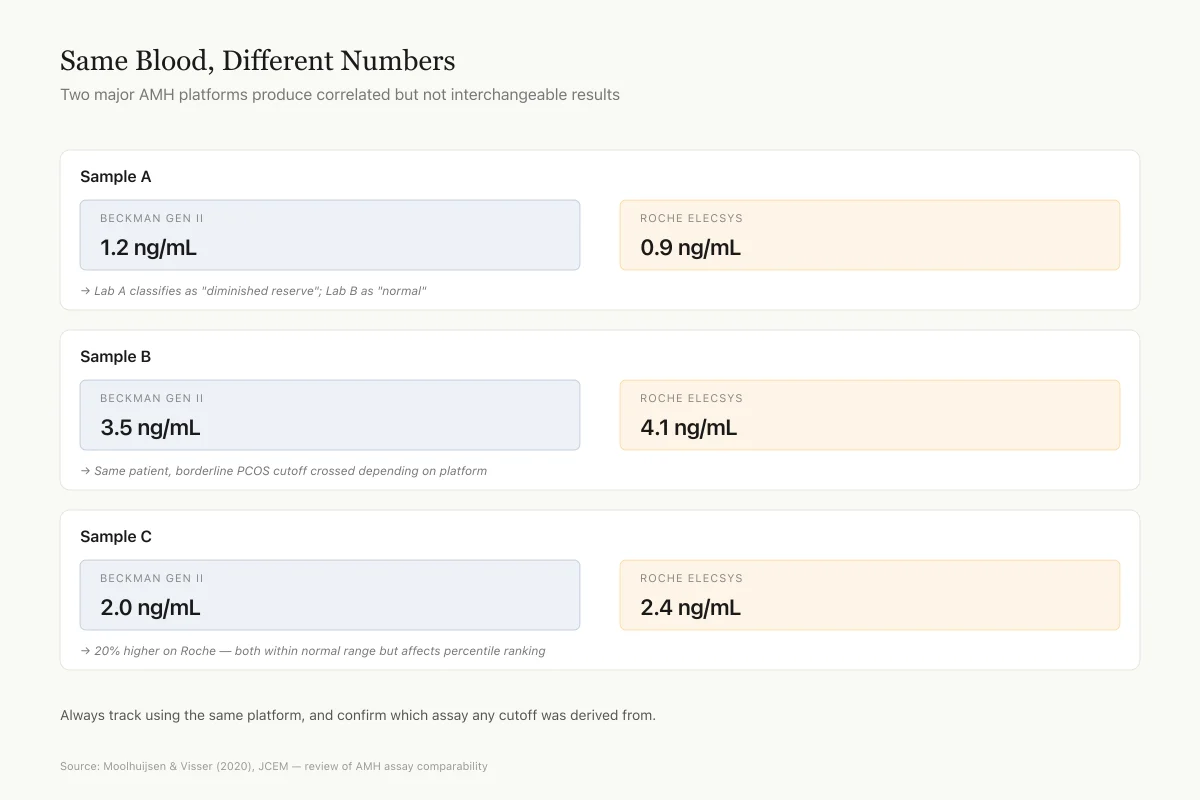
The Standardization Problem
Same Blood, Different Numbers
Two major AMH platforms produce correlated but not interchangeable results
Sample A
Beckman Gen II
1.2 ng/mL
Roche Elecsys
0.9 ng/mL
→ Lab A classifies as "diminished reserve"; Lab B as "normal"
Sample B
Beckman Gen II
3.5 ng/mL
Roche Elecsys
4.1 ng/mL
→ Same patient, borderline PCOS cutoff crossed depending on platform
Sample C
Beckman Gen II
2 ng/mL
Roche Elecsys
2.4 ng/mL
→ 20% higher on Roche — both within normal range but affects percentile ranking
Published cutoffs for "diminished reserve," "PCOS diagnosis," and "IVF dosing" are platform-specific. A 2.0 ng/mL result from one lab may not mean the same thing as 2.0 from another. Always track using the same platform, and confirm which assay any cutoff was derived from.
Source: Moolhuijsen & Visser (2020), JCEM — review of AMH assay comparability
 View as image
View as imageLimitations of AMH as a Biomarker
Quantity, not quality: AMH measures how many follicles remain, not whether those follicles contain chromosomally normal eggs. Age remains the strongest predictor of egg quality and pregnancy success.
Not a fertility test: A normal AMH does not guarantee fertility. A low AMH does not mean infertility. AMH predicts ovarian response to stimulation, not the ability to conceive naturally.
Assay variability: Different AMH assays give different values for the same sample. Published cutoffs are platform-specific, and a universal reference standard does not yet exist.
Population differences: AMH reference ranges were developed primarily in European and North American populations. Ethnicity, BMI, and vitamin D status can all influence AMH levels, and population-specific reference ranges are still being established.
Not validated for general screening: Professional societies recommend against using AMH to counsel women not seeking fertility treatment. The test was designed for reproductive medicine, not general health screening.
Hormonal contraception: Oral contraceptive pills suppress AMH levels by 20-30%, which can lead to falsely low results in women using hormonal birth control.
The Bottom Line
Anti-Mullerian hormone is a 140 kDa glycoprotein produced by ovarian granulosa cells that serves as the best available blood marker of ovarian reserve. It outperforms FSH for predicting IVF response, can be measured on any day of the menstrual cycle, and declines progressively toward menopause. AMH is elevated 2-3 fold in PCOS and modestly improves menopause timing predictions. Its primary limitation is that it measures follicle quantity, not egg quality. Assay standardization remains an unresolved challenge that complicates clinical interpretation.
Sources & References
- 1RPEP-02339·Broer, Simone L et al. (2014). “Anti-Müllerian hormone: ovarian reserve testing and its potential clinical implications..” Human reproduction update.Study breakdown →PubMed →↩
- 2RPEP-02370·Dewailly, Didier et al. (2014). “The physiology and clinical utility of anti-Mullerian hormone in women..” Human reproduction update.Study breakdown →PubMed →↩
- 4RPEP-05007·Moolhuijsen, Loes M E et al. (2020). “Anti-Müllerian Hormone: What This Fertility Blood Test Can and Cannot Tell You.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 5RPEP-01643·La Marca, A et al. (2010). “Anti-Mullerian hormone (AMH) as a predictive marker in assisted reproductive technology (ART)..” Human reproduction update.Study breakdown →PubMed →↩
- 6RPEP-01797·La Marca, A et al. (2011). “AMH Blood Test Helps Predict IVF Success When Combined with Age.” Reproductive biomedicine online.Study breakdown →PubMed →↩
- 7RPEP-04203·Granger, Emily et al. (2019). “Anti-Müllerian Hormone and Its Predictive Utility in Assisted Reproductive Technologies Outcomes..” Clinical obstetrics and gynecology.Study breakdown →PubMed →↩
- 8RPEP-02876·Bhide, Priya et al. (2016). “Anti-Müllerian hormone and polycystic ovary syndrome..” Best practice & research. Clinical obstetrics & gynaecology.Study breakdown →PubMed →↩
- 9RPEP-07220·Nelson, Scott M et al. (2023). “Anti-Müllerian hormone for the diagnosis and prediction of menopause: a systematic review..” Human reproduction update.Study breakdown →PubMed →↩
- 10RPEP-06466·Russell, Nicole et al. (2022). “Anti-Müllerian Hormone: The Peptide Biomarker That Predicts Ovarian Reserve and Fertility.” Journal of clinical medicine.Study breakdown →PubMed →↩